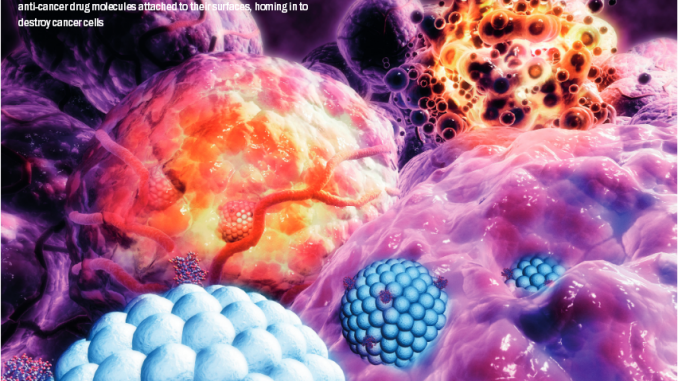
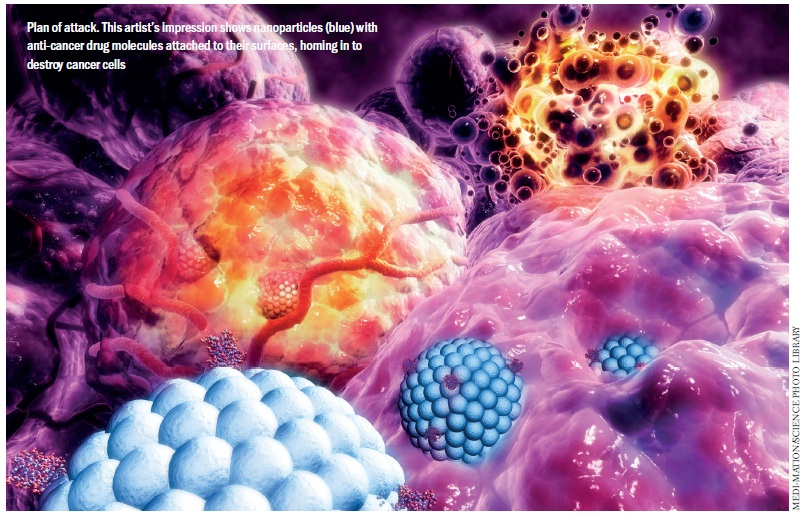
It’s long been the stuff of science fiction, but could the prospect of nanotechnology systems that deliver atomic bombs’ direct to cancer cells soon become reality?
It’s a compelling vision: tiny particles, primed with a highly effective cancer killing drug, able to move round the body and seek out and destroy tumour cells, only releasing their deadly payload when they reach their target. That’s precisely what many research teams are now developing with nanotechnology, a rapidly growing field concerned with the properties of materials at the nanometre scale – particles of 10-9 metres, a size that is bigger than molecules but smaller than human cells (if a nanoparticle were a football, a red blood cell would be the size of a football field).
If indeed nanoparticles can find their targets they could open up a new era of cancer treatment that avoids several big obstacles with usual drug therapy. By holding toxic chemotherapies inside shells made from substances such as polymers, the rest of the body can be protected from harmful side-effects, and drugs can circulate for longer, with more finding the target. Drugs that have been too toxic to use before are now making a reappearance in nano form, and to help them find the target, nano-particles are also being designed with specific tumour-seeking agents, in a further development of the field of targeted biological therapy.
Combining primary targeting (getting to where tumours are, and even chasing down circulating cancer cells) with secondary targeting (attacking cancer cells and structures inside the cell) is what is making nanotechnology potentially so exciting.
And that’s only part of the story. Other therapies in development don’t involve chemotherapy at all. Some, for example deliver pieces of genetic materials to turn off genes implicated in cancer cell growth; others concentrate metallic particles that can kill tumours when external radiation such as heat is applied. Nanoparticles can also be injected directly into tumours, or even by inhaler, for delivering lung cancer therapies direct to the tumours.
There is also a rapidly growing interest in the use of nanotechnology in diagnostics. It is the therapeutic use of nanotechnology that has captured the most headlines, however, especially as the main applications in development are for treating advanced disease in patients for whom other approaches have had little success.
Exploiting an Achilles heel
Key to the potential of nanomedicine in cancer is a crucial difference between healthy and cancerous cells that these techniques can exploit. While cancer is notorious for its ability to evade treatment, at the nanoscale – at least on a mechanical basis – solid tumours have a fundamental weakness, as Jennifer Grossman, a scientist at the Nanotechnology Characterization Laboratory in the US, part of the National Cancer Institute (NCI), explains. “We have known for some time that particulate matter can accumulate in inflammatory sites such as tumours. Inflamed tissues have a different vasculature than healthy tissue because the blood vessels grow very quickly and irregularly and have pores or holes that are big enough for a nanoparticle to pass inside – but the particles are too big to get through the wall of a healthy blood vessel. So when you inject them into the bloodstream they stay there except through these pores in tumour vasculature.”
That is simplified description. The scientific term for this is the enhanced permeability and retention (EPR) effect, where retention refers to the lack of lymphatics in tumours, which helps to stop particles draining away quickly. It has only been verified in animal models, she says, and while all human tumours have ‘leaky’ vasculature, this varies greatly depending on the cancer type. The effect also depends on tumour size, and small tumours may lack enough vasculature to be targeted.
The difference in size of the openings between healthy and tumour tissue can be huge – just 2–4 nanometres for healthy tissue and several hundred nanometres for cancer blood vessels. With the US putting its weight behind nanotechnology (the 21st century Nanotechnology Research and Development Act came in 2003), researchers – and in particular chemical engineers and material scientists – have since helped to determine the optimum size and other properties for nanoparticles, such that they have a good chance of reaching tumours.
Too small, and like small molecule drugs they can be filtered out in the kidneys unless bound to blood proteins; too large, and they may stay in blood vessels and not penetrate a tumour, and they also need to evade the body’s defence system, which carries foreign bodies such as viruses to the spleen and liver (although the latter is itself a common site for metastases and a target itself).
As Mark Davis, a chemical engineer at Caltech (California Institute of Technology) has reported, it turns out that the limits for particles are 10–100 nm, with 30–70 nm being about optimum. Davis became a pioneer in cancer nanoparticles after his wife was diagnosed with the disease. With others he has gone on to determine more design features that are important, as well as experimenting with a huge array of possible materials and structures that could optimise delivery of drugs and other agents such as gene silencing with fragments of RNA. Because nanoparticles have the special property of a large surface area relative to volume, researchers have put much emphasis on coatings that can both pave the way to the tumour and engage with it.
Overcoming barriers
Grossman details a range of factors that stand in the way of particles even getting to a tumour, despite the attraction of the EPR effect. First, there is the body’s phagocyte system, which protects against viruses and other foreign bodies. The chances of being carried away by this can be reduced by applying a coating such as polyethylene glycol (PEG), which she says also adjusts the electrical charge of the particle surface to make it less likely to be attracted to healthy cells before it reaches a tumour.
Then the tumour itself has its own protective shell – the stroma – which is made up of a number of structures including a collagen matrix, which Grossman notes can be very dense in some tumours such as pancreatic, and which can stand in the way of drugs (and is one reason for the poor outlook for these cancers). Tumours can also have a high fluid pressure because they lack drainage, which can impede the inflow of particles. And if nano-drugs only reach the edges of tumours and have limited effect, then drug resistance could build up. It’s no wonder that new fields such as ‘transport oncophysics’ are springing up to help find ways around the barriers.
Apart from the design of particles (such as disguising them as red blood cells, which some groups are doing) there are many approaches to aiding their passage, such as using antiangiogenic drugs to lower pressures in tumours.
With several functions to perform, it can be hard to build these particles with the necessary quality and purity in sufficient quantities for large-scale use. Adding more complexity can also adversely affect desirable properties such as the ability to circulate long enough in the bloodstream, though the opposite can also pose a problem: nano-drugs combining a carrying device as well as drugs can be difficult to eliminate from the body.
Questions remain about whether cancer nanotechology has too many such barriers to move ahead. Development of new therapeutics and diagnostics is still mostly in the hands of academic and government research labs and start-up biotech companies, particularly in high-tech ‘clusters’ such as around Boston in the US. However, large pharmaceutical companies have recently struck deals with some of the start-ups, indicating a step change in investment and potential, and a number of new nano-drugs have reached phase II and III stages.
This is a field that is also bringing together different disciplines, which cancer visionaries feel will be crucial, in one way or another, to making advances – physicists, engineers, material scientists, computing experts and chemists are among those making the running in cancer nanotechnology, alongside colleagues in biology and the clinic. The combined brain power now focusing on cancer nanotechnology is formidable, while other branches of nanomedicine are also emerging.
In the clinic
The first nano-drugs use the passive targeting of the EPR effect to accumulate in tumours and diffuse their payload; much of the new generation are taking an active approach by using targeted coatings such as antibodies, proteins and peptides that bind to tumour cells and only then release a drug that diffuses in the tumour, or enters tumour cells.
For drug delivery to tumours, the first application – Doxil, or Caelyx in Europe – was approved as long ago as 1995 by the US FDA and is approved for recurrent ovarian cancer, relapsed multiple myeloma, metastatic breast cancer and Kaposi’s sarcoma. Now out of patent, today’s biotechnology experts consider it to be very much a first-generation nanoparticle, as it is not on the same small scale or ‘smartness’ as systems now being engineered, and relies only on passive targeting.
Doxil is made from a liposome, which comprises a fatty sphere surrounding a core that contains doxo-rubicin – a chemotherapy with dose-limiting toxicity. It also has a polymer coating that allows it to stay in the bloodstream longer (but can cause skin side-effects). A similar nano-drug, Myocet, does not have the coating, and is approved in Europe and Canada for treating patients with metastatic breast cancer. The benefit of both these nano-drugs when used in breast cancer lies in their reduced heart toxicity, as there is no reported gain in efficacy. This nano-approach set in train the search for ways to deliver therapies that would be too toxic to be approved for more systemic administration – both new and existing drugs that may have been abandoned or denied approval on safety grounds in the past.
Another nano-drug in use is Abraxane, which was approved more recently in the US (2005) for metastatic breast cancer, and raised hopes that it would finally usher in a lot more agents. It is formulated to overcome a different type of toxicity, and has also been found to be more effective than the conventional drug at second line or more – in this case paclitaxel (Taxol), which is insoluble and so is prepared with a solvent that can cause side effects. Abraxane attaches paclitaxel to nanoparticles made from the human protein, albumin. A Japanese company, NanoCarrier, currently has a phase III trial of paclitaxel using nanoparticles made instead of polymeric micelles, which are also able to carry insoluble drugs.
There are a few other approved nano-drugs – a recent one that gained accelerated approval from the FDA is Marqibo, a liposomal formulation of the cancer drug vincristine, for patients with relapsed acute lymphoblastic leukaemia that is negative for the Philadelphia chromosome.
In the pipeline
While other potential nano-drugs have investigational approval, the momentum depends on pushing the boundaries from the first-generation products and gaining investment from mainstream drug development. As Elisabet de los Pinos, chief executive of Aura Biosciences, one of the biotech firms in the Boston cluster, says, nanotechnology involves a change of mindset – not drug discovery but better drug delivery for the large number of drugs already known to be effective. “While we have some phenomenal new targeted agents such as monoclonal antibodies, they are often given in combination with very toxic drugs such as the platinums, which are still used in every lung cancer patient. We need to deliver a targeted ‘atomic bomb’ directly to the tumour.”
 Viral delivery
Viral delivery
The technology that Aura Biosciences is using is based on viruses, which could better deliver treatments by combining small size and more precise targeting. “We are piggybacking on nature with viruses, because they are below 100 nanometres and already penetrate the body’s barriers,” says de los Pinos. “But you also need to better distinguish between normal and tumour cells, so you need a targeting method – and technologies based on materials such as poly-mers and carbon structures have a problem when you want to deliver a toxic agent. You need to be specific in where it’s delivered, otherwise you won’t get approval to use it.”
The virus particles that Aura is building are called pseudovirions, which are synthetic viruses but without any viral DNA – they are simply protein shells – and a group at the NCI has found they possess a critical property of ‘infecting’ tumour cells but not normal ones. De los Pinos, a Spaniard whose background is molecular biology, says making use of this natural propensity to target tumour cells is the unique approach her company is taking, and that she was set on virus technology from the start of her venture, looking at various approaches being developed in Europe and the US, opting eventually for technologies from France and the US, and also then establishing the company in the Boston hotspot.
“Like most biotech firms we have great technology from top academic centres, but you have to scale it so we have something that is usable in clinical trials, which is what we have been working on for the last year or so. It’s not straightforward because the technology is novel and we are the only ones doing it, but we are ready now to get the approval from the FDA to dose a first patient.”
De los Pinos won’t disclose the treatment that could go into a first human trial with Aura’s pseudovirions, but animal models have demonstrated uptake in metastatic ovarian cancer and non-small-cell lung cancer. Aura has some heavyweight backing, including José Baselga, the Spanish medical oncologist and former ESMO president, who is now chief physician at the Memorial Sloan-Kettering Cancer Center in New York, and is a member of the company’s board of directors.
Ligand loaded polymers
On Aura’s doorstep in Boston are other companies focused on new ways to deliver anti-cancer therapies, and some have moved into clinical trials – with varying success so far. Bind Therapeutics, which was co-founded by Robert Langer, a biotech pioneer and chemical engineer at MIT, has polymer-based particles it calls Accurins, which achieve targeting with ligand molecules attached to the particle surface. So far, the company has completed a phase I study for its BIND-014 agent, which is an Accurin containing docetaxel and which targets the prostate-specific membrane antigen (PSMA), a cell-surface protein that the company says is abundantly expressed on certain cancer cells and on new blood vessels that feed a wide array of solid tumours.
Bind’s technology has certainly impressed several large drug companies: Amgen, AstraZeneca and Pfizer have signed up to deals worth several hundred million dollars in total for nano-delivery of agents that are yet to be specified, but will include targeted drugs such as kinase inhibitors.
Greater toxicity more safely
Meanwhile Cerulean Pharma, with which Caltech’s Mark Davis is involved, had progressed to a phase II trial for its lead candidate, CRLX101, which carries camptothecin – a highly potent anti-cancer agent that was discovered in the 1960s but only used in less potent derivatives, namely irinotecan and topotecan. Cerulean’s technology uses cyclodextrins, made up of sugar molecules, linked to a polymer, to create nanoparticles with a slow-release mechanism to target tumour cells. However, the phase II study, conducted in more than 150 patients with advanced lung cancer in Russia and the Ukraine, did not show survival benefit – a setback for the company, though it does have other ongoing trials and technology.
A liposome nano-drug containing irinotecan has reached phase III – Merrimack Pharmaceuticals is running the NAPOLI-1 study for second-line metastatic pancreatic cancer patients. The particle relies on the natural blood flow of the tumour to direct the therapy to the cancer.
The many possibilities of metals
Metals are another class of material that are being used for therapeutic applications, although Grossman says that, as they do not degrade, there are concerns about safety if they persist in the body. But metal particles are a major avenue for researchers and biotech firms. CytImmune Sciences, for example, another firm in the Boston area, has completed a phase I trial of gold nanoparticles carrying tumour necrosis factor (TNF), a toxic agent that targets tumour vasculature. Gold has properties that make it especially useful at the nano-level, but other metals are being used.
Ian Baker, a materials scientist and director of the Dartmouth Center for Cancer Nanotechnology Excellence in the US, says that among his projects are investigations of iron nanoparticles that, once located in tumours, can be heated to destroy cells – in this case by using a technique called magnetic hyperthermia, where a magnetic field heats iron oxide particles. “The idea of hyperthermia has been around for long time, but the problem is that tumour cells don’t respond differently to heat than normal cells, so we need to inject the particles locally or tag them with antibodies so they find their way to the tumour. Mostly what we are doing is heating, but one project is looking at heating to release a drug. We can cure cancer in mice with hyperthermia, and are looking to do clinical trials, but there are more restrictions in the US.”
A team in Germany, he notes, has been working on magnetic hyperthermia for more than 20 years; a company called MagForce has so far taken the technology to a phase II trial in grade IV brain tumours (glioblastoma multiforme) and a phase I in pancreatic and prostate tumours. Apart from destroying the cells by heating, or thermal ablation, the particles are also said to sensitise tumours to chemo- or radiotherapy. Clearly there could be important crossovers here with other disciplines such as interventional radiology and radiation oncology: another example is French firm Nanobiotix, whose NanoXray particles enhance radiotherapy.
Gene silencing
Potentially one of the most elegant uses for therapeutic nanoparticles is to carry segments of RNA that can directly turn off genes in cancer cells, and so bypass the expressed proteins that are the usual targets of drugs – if they can be targeted at all. A number of research groups are involved with siRNA – small interfering RNA molecules – which are delivered by nanoparticles, as RNA does not survive for long on its own after injection into the bloodstream. There are a number of trials at phase I/II aimed at silencing various protein expressions, such as from the aptly named British firm, Silence Therapeutics, Alnylam Pharmaceuticals and Arrowhead Research in the US, the latter a majority owner of a firm that Mark Davis founded and which was the first to demonstrate that a targeted nanoparticle could deliver siRNA in a human cancer patient.
It’s even possible to assemble these RNA molecules into a nano-structure built from DNA, which researchers at MIT in the US have done, showing that the resulting particles survive long enough in animal models to reach tumours.
DNA origami
Björn Högberg, a principal investigator at the Swedish Medical Nanoscience Centre at the Karolinska Institute, Sweden, is also researching DNA. “We don’t care about the genetics but see DNA as a building material for nanoparticles in different shapes – circles, rods, crosses, smiley faces – anything we want. It’s called DNA origami and is like 3D printing technology for nanoscale objects, and we can design one in just a few days and get a high yield of particles.”
In published work, Högberg and his team have designed a DNA nanoparticle that carries the chemotherapy drug doxorubicin, which “likes to attach to DNA”, he says “We can attach molecules like this and know exactly where they will sit. Other nanoparticle technologies don’t have this perfect positional control.” This is early work, he adds, but the way it would work is that doxorubicin would better diffuse to a tumour cell’s DNA and block cell reproduction.

A nano-boost for Europe?
The US probably has a lead in cancer nanotechnology, thanks to the NCI Alliance for Nanotechnology, which funds ‘centres of excellence’ such as Baker’s Dartmouth Center for Cancer Nanotechnology Excellence, and also product partnerships and training centres, although Baker says the funding does not extend to clinical trials.
Grossman’s facility – the Characterization Lab – provides a free service for biotech companies to test the properties of nano-formulations, including animal testing, for safety and efficacy, and is a collaboration between the NCI, FDA and the US National Institute of Standards and Technology. This service is a particular strength, she says, because of the complexity of nano-products. “There’s a natural heterogeneity for nanoparticles, as they are made of thousands of atoms, so we need to characterise their purity and quality – it’s more complicated than making a small molecule drug.” More than 300 different nano-formulations developed by over 75 research groups have been ‘characterised’ so far, and at least six are in clinical trials.
Grossman says officials at the European Commission are interested in setting up a similar characterisation capability in Europe, and to that end she’s recently had a meeting in Brussels to highlight the work of the NCI lab, which could be good news for the growing number of biotech firms on this side of the Atlantic. An announcement about work around establishing an EU nanotechnology characterisation facility, and collaboration with the US, was expected from the European Technology Platform on Nanomedicine (ETPN) – a high-level group of industry and academic scientists – at the European Summit on Clinical Nanomedicine in Basel in late June.
Last February saw the launch of the European School of DNA Nanotechnology, an Initial Training Network under the EU’s 7th framework programme, which is set to run for four years. A joint initiative by leading scientists from academic centres in Denmark, Germany, Sweden (Karolinska Institute, where Högberg is based) and the UK, which also involves commercial companies, the School aims to train early-stage researchers specifically in the interdisciplinary field of DNA nanotechnology, and to promote the foundation of new bionanotechnology start-up companies.
The question is whether all this will be enough to persuade pioneers like Elisabet de los Pinos, that Europe can offer the supportive environment needed for success.



