A chromosomal alteration present in almost all patients with Hodgkin lymphoma makes the disease uniquely vulnerable to PD-1/PD-L1 immune checkpoint blockers. Astrid Pavlovsky reviews the trial evidence and clinical experience, and looks to the future possible use of this class of therapy.
This grandround was first presented by Astrid Pavlovsky, from the Department of Haematology, Fundaleu, Buenos Aires, as a live webcast for the European School of Oncology. Emmanuele Zucca, from the Oncology Institute of Southern Switzerland, Bellinzona, posed questions raised during the presentation. It was edited by Susan Mayor. The webcast of this and other e-sessions can be accessed at e-eso.net.
The treatment of Hodgkin lymphoma is a success story in haemato-oncology, with most patients, whether at an early or advanced stage of the disease, being cured with first-line treatment. However, 25–35% of patients have primary refractory or relapsed Hodgkin lymphoma, and a proportion will eventually die of it. We have known for more than a decade that the standard-of-care for patients who relapse after first-line treatment is salvage chemotherapy and autologous stem cell transplantation (ASCT). Two clinical trials have shown that ASCT is associated with significantly greater freedom from treatment failure, which is achieved in around 50% of patients, so this is our first choice of treatment for these patients.
Unfortunately, this means that about 50% of patients relapse after ASCT, and the post-progression survival for this group is poor. Up until 2013, patients who relapsed in the first year after transplant had a median survival of only about one year from the start of disease progression, and for those who relapsed after the first year following transplant, median post-progression survival was only around two years.
Salvage chemotherapy after failure of ASCT has not shown very promising results, and controls disease for only seven to ten months, so this is a subgroup of patients where, until recently, there has been no good standard of care treatment. Different salvage chemotherapy agents, including gemcitabine and vinblastine, have been tried, but with low overall and complete response rates, and with accompanying haematological toxicity.
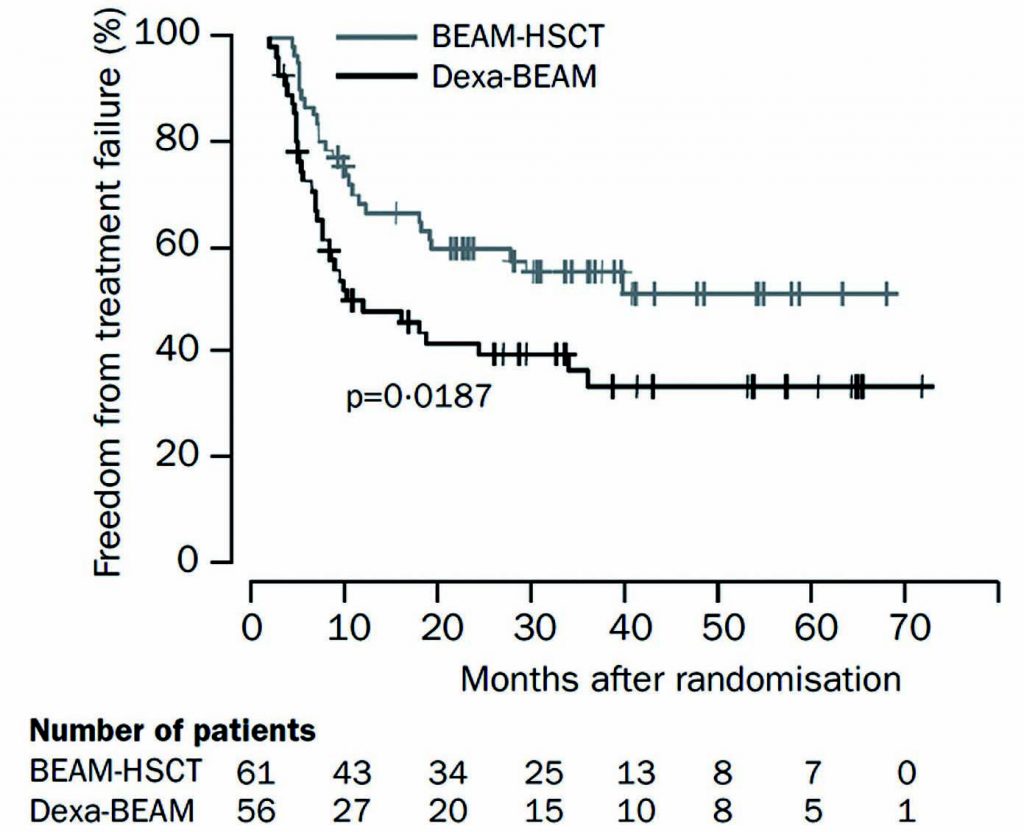
Source: N Schmitz et al. (2002) Lancet 359:2065–71, reprinted with permission from Elsevier
Recently, we have seen very impressive results with monotherapy for the first time, with the use of brentuximab vedotin in patients who have failed after ASCT, with an overall response rate of 75% and a 34% complete response rate (JCO 2012, 30:2183–89). However, very few of these patients maintain complete remission, and they eventually relapse.
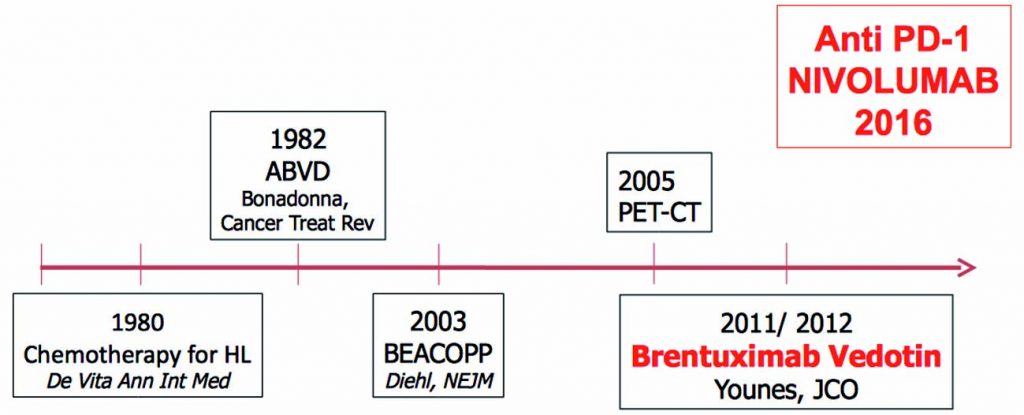
The figure below traces progress in the management of Hodgkin lymphoma over the last few decades, from the introduction of chemotherapy in 1980, with the MOPP regimen (Ann Intern Med 1980, 92:587–95), followed swiftly by the ABVD regimen in 1982 (Cancer Treat Rev 1982, 9:21–35). More recently, BEACOPP was introduced in 2003 (NEJM 2003, 348:2386–95), followed by the introduction of brentuximab vedotin in 2012 (JCO 2012, 30:2183–89). Nivolumab became available in 2016 for use in patients who have relapsed after ASCT.
Harnessing the power of the immune system
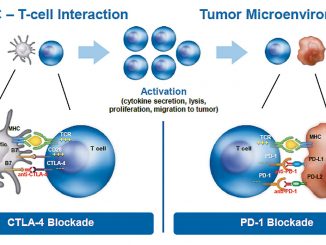
For decades, we have been trying to harness the power of the body’s immune system to combat tumour cell growth. Hodgkin lymphoma is a clear example of a disease where there are only a few malignant cells together with a very extensive immune infiltrate.
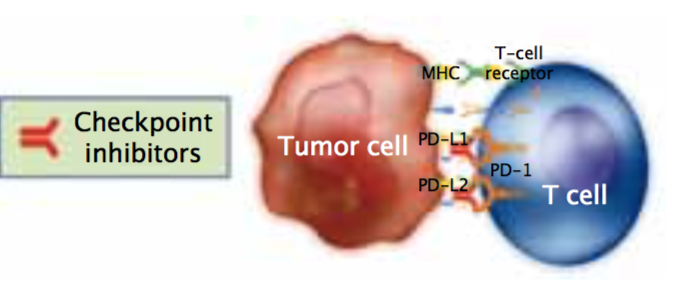
However, this immune infiltrate is ineffective because the tumour cells are still able to grow. So, how can we optimise the power of the immune system to become a line of therapy? Programmed cell death ligand 1 (PD-L1) is an immunomodulatory molecule expressed by antigen-presenting cells as well as by certain tumour cells. It binds to T cell receptors, thereby inhibiting T-cell-mediated immunity.
Almost all patients with Hodgkin lymphoma have an alteration in chromosome 9p24, causing an over-expression of PD-L1 and PD-L2 on the surface of Reed-Sternberg cells, which leads to immune evasion.
This over-expression makes Hodgkin lymphoma uniquely vulnerable to PD-L1 blockade, as it appears that Hodgkin lymphoma cells depend on this mechanism to survive.
In addition, the 9p24 amplification leads to over-expression of JAK-2, and this mechanism is also being investigated in immuno-oncology.
Data on PD-L1 expression in Hodgkin lymphoma
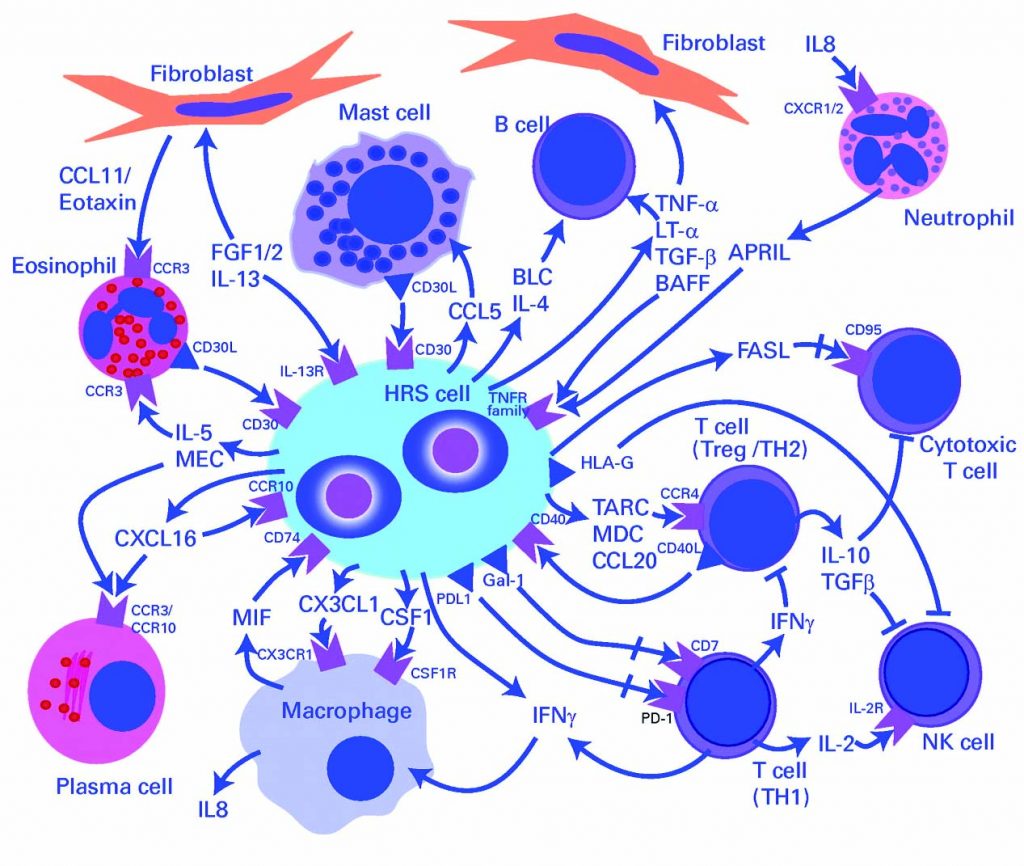
HRS – Hodgkin and Reed-Sternberg cells]
A recent study of diagnostic biopsies in 108 patients treated with ABVD showed that all had 9p24 genetic alterations. These alterations were either disomies, polysomies, amplification, copy gain or translocations. Progression free survival (PFS) of patients was stratified according to stage. Three prognostic groups were seen: early favourable, early unfavourable and advanced stage. In a parallel analysis, PFS of patients was stratified according to type of 9p24 alteration. These resulted in new prognostic groups, showing that the five patients who had polysomy had 100% PFS, and patients with amplification of 9p24 had the worst PFS (JCO 2016, 34:2690–97). The prognosis based on clinical stage may be linked to the prevalence of 9p24 alterations. Only 24% of patients with an early stage favourable prognosis had 9p24 amplification, compared to 34% of those with early stage unfavourable prognosis and 50% of those with advanced stage disease. This is the first time we have had a pre-treatment genetic predictor of prognosis in patients with Hodgkin lymphoma (see figure).
A further study analysed the prognostic impact of PD-1 expression in tumour infiltrating leukocytes in diagnostic Hodgkin lymphoma biopsies in 415 patients treated with ABVD, with or without radiotherapy (ISHL 2016, T005). They were divided into either low PD-1 expression (<10% PD-1 positive leukocytes, 85% of patients) or high expression (>10% PD-1 positive leukocytes, 15% of patients). Patients in the high expression group showed an inferior event-free survival compared with the low-expression group. This gives a further possible prognostic tool: patients with high-expression of PD-1 might benefit more from high-intensity chemotherapy or a PD-1 inhibitor as first-line therapy.
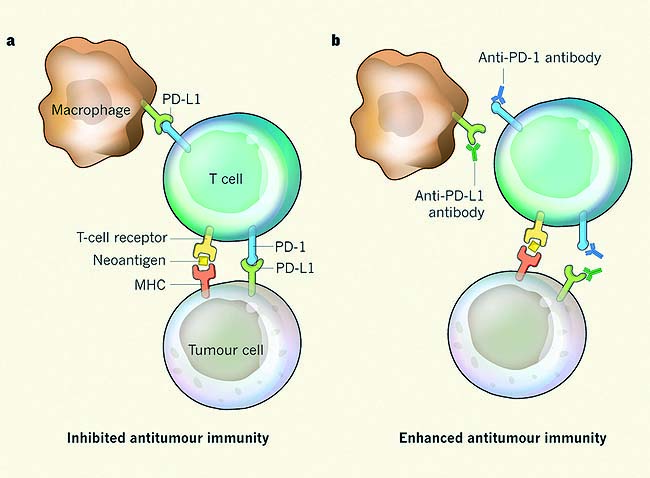
It binds to T cell receptors, helping tumour cells evade detection by the immune system
These new findings may in the future have implications for planning upfront therapy in Hodgkin lymphoma, recognising that advanced stage disease is associated with inferior outcome and that 9p24 amplification is also associated with an unfavourable outcome. Amplification of 9p24 is more common in advanced stage disease, and Hodgkin lymphoma patients with high PD-1 expression have inferior event-free survival after ABVD compared to patients with low expression.
Studies with immune checkpoint blockade therapy in Hodgkins
We now have two drugs – nivolumab and pembrolizumab – that are fully humanised monoclonal antibodies that block interaction between PD1 and PD-L1, which inhibits T cell activity to restore normal immunological function.
A phase I study (CA209-039) investigated nivolumab in 105 patients with relapsed or refractory lymphoid malignancies and classical Hodgkin lymphoma. Exclusion criteria were: no existing autoimmune disease, no prior organ or stem cell transplant and no prior checkpoint blockade. Of the 23 patients with Hodgkin lymphoma, 78% had undergone prior ASCT and 78% had received prior brentuximab therapy and had failed both, so consequently had poor prognosis.
Results showed that, of all patients with haematological malignancies, patients with Hodgkin lymphoma had the best response to nivolumab, with an overall response rate of 87%, including 26% with complete response and 50% with ongoing response to treatment on follow-up (median 74 weeks). Nivolumab continued until progression or unacceptable toxicity (Timmerman et al, 2015, 13th International Conference on Malignant Lymphoma, Lugano, Abstract #010).
The figure left shows a spider plot of responses to nivolumab, demonstrating the wide variation in response to treatment in Hodgkin lymphoma patients. One patient’s lesion grew markedly following treatment before showing a partial response. In contrast, two patients had first complete remissions within four to six weeks. After 76 weeks of treatment, most patients had a sustained partial response. Further analysis showed that two patients maintained a complete response even after stopping treatment, and two patients had late complete responses, with one occurring after stopping therapy.
These findings show that the immunological response to nivolumab is quite variable and individual for each patient. It is very different to that seen with chemotherapy, so we have to evaluate response differently.
It is important to recognise that some individuals have early progression of their lesions followed by subsequent remission, others have durable partial responses with clinical benefit, some patients have late complete responses or improvement in response after stopping treatment, and others have new lesions with or without reduction of pre-existing lesions followed by late response to treatment.
Longer-term follow-up showed that that progression-free survival was 50% after a median follow-up period of 92 weeks, and overall survival was 74%. This is promising and represents a new treatment option for patients with aggressive Hodgkin lymphoma. When the data were reported, most patients had an ongoing response, so further evaluation is ongoing.
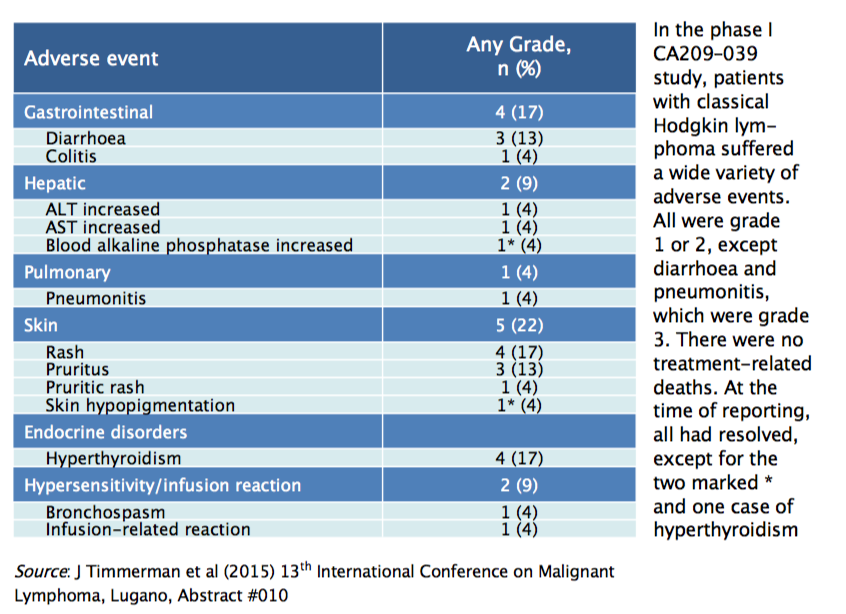
Drug-related adverse events were relatively frequent, with 83% of Hodgkin lymphoma patients having some kind of adverse event, but severe adverse events were quite rare. All adverse events were grade 1 or 2 except diarrhoea and pneumonitis, which were grade 3. There were no grade 4 or 5 events nor deaths related to treatment; 14% of patients discontinued nivolumab treatment due to drug-related adverse events. Most of the adverse events were haematological or skin-related (see table), and the majority occurred in the first few weeks of treatment, before resolving. However, some patients had severe adverse events later in treatment, so they must be closely monitored using an appropriate algorithm to distinguish between immune-related adverse events and disease progression, to determine subsequent management. Almost all adverse events resolved with management, except for hyperthyroidism, which should be managed by a specialist.
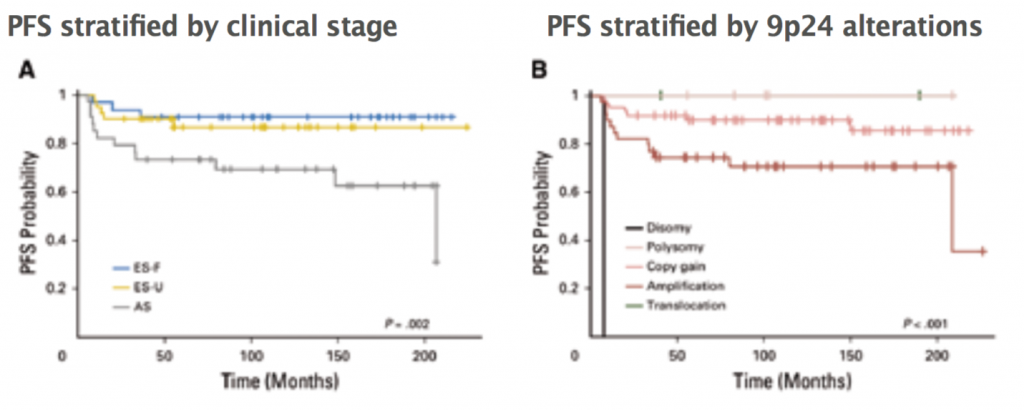
A: ES-F – early stage favourable (n=33), ES-U – early stage unfavourable (n=41), AS – advanced stage (n=34); P=0.002 log-rank test. B: Disomy (n=1), polysomy (n=5), copy gain (n=61), amplification (n=39), translocation (n=2); P<0.001 log-rank test
Source: MG Roemer et al (2016) JCO 34:2690–97, reprinted with permission from the American Society of Clinical Oncology. All rights reserved
A further phase II study (CheckMate 205) investigated nivolumab in Hodgkin lymphoma patients divided into three cohorts – patients who had failed after ASCT (cohort A), patients who had undergone ASCT followed by brentuximab which then failed (cohort B), and patients who had received ASCT after or before brentuximab (cohort C). Focussing on cohort B, which included patients who had previously failed ASCT and brentuximab therapy, the patients were given 3 mg/kg nivolumab every two weeks until disease progression or unacceptable toxicity. Most patients were treated as outpatients, as nivolumab is a very easy infusion to administer. The objective response rate was 66%, with 9% of patients achieving complete remission. The median time to response was two months, with a median duration of response of eight months. Overall survival at six months was 98% and progression-free survival was 77%, with a median progression-free survival of ten months (Engert et al, oral presentation, European Hematology Association, June 2016).
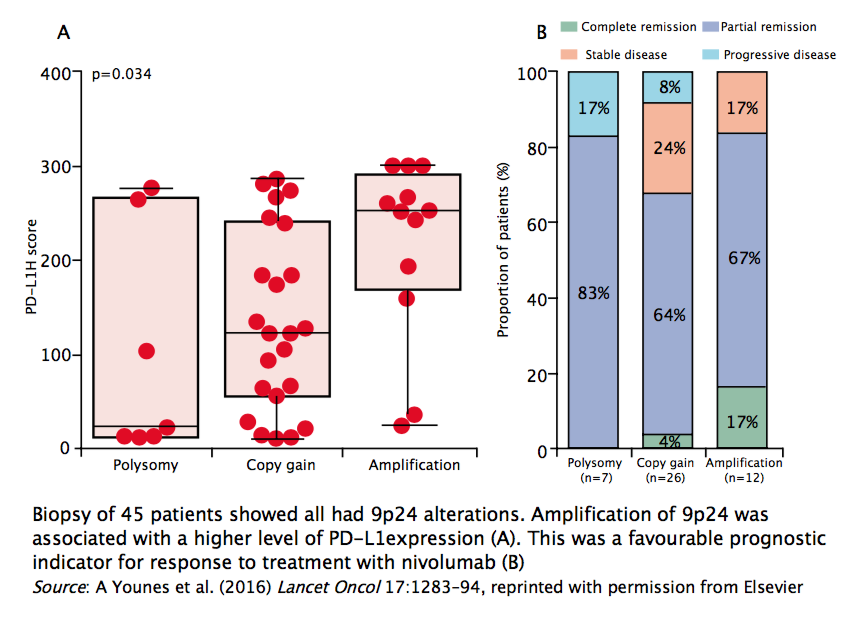
Forty-five patients were analysed for genetic alterations, and all showed 9p24 alterations, including 85% with either copy gain or amplification. Responses were observed in patients with all types of genetic alteration at 9p24. Patients with amplification of 9p24 had a higher level of PD-L1 expression. This was a favourable prognostic indicator for response to treatment with nivolumab, with 17% showing complete response, which was higher than in the other two groups. In this trial, higher levels of PD-L1 expression associated with 9p24 amplification were associated with greater response to nivolumab with better treatment outcomes.
Defining response to immunotherapy in Hodgkin lymphoma
Until now we have used the Lugano Classification to assess response to treatment, particularly for chemotherapy, incorporating PET to evaluate patients with Hodgkin lymphoma. Chesson and colleagues have published a refinement of the Lugano Classification that takes into account the findings on response to immunotherapy: the Lymphoma Response to Immunomodulatory Therapy Criteria (LyRIC, Blood 2016, 128:2489–98).
These criteria include an indeterminate response (IR), which is a provisional term to identify lesions that may be flares or pseudoprogression of disease triggered by an inflammatory response to therapy, in contrast to indicating progressive disease. In the future, we should hopefully be able to distinguish between these two mechanisms but, for now, the definition allows appropriate patients to remain on treatment until reassessment can accurately confirm or refute progressive disease. There are three classifications of indeterminate response (IR):
IR1 refers to an increase in tumour size of 50% or greater in the first 12 weeks of treatment, but with no clinical progression.
IR2 indicates the appearance of a new lesion or growth of one or more existing lesions of 50% or greater at any time during treatment, in the absence of overall progression of tumour burden.
IR3 refers to an increase in PET-FDG uptake (metabolic activity) of one or more lesions with no increase in lesion size or number.
The expert panel recommends repeat scanning after 12 weeks and carrying out a further biopsy to gain a clearer idea of whether a patient has progressive disease or not.
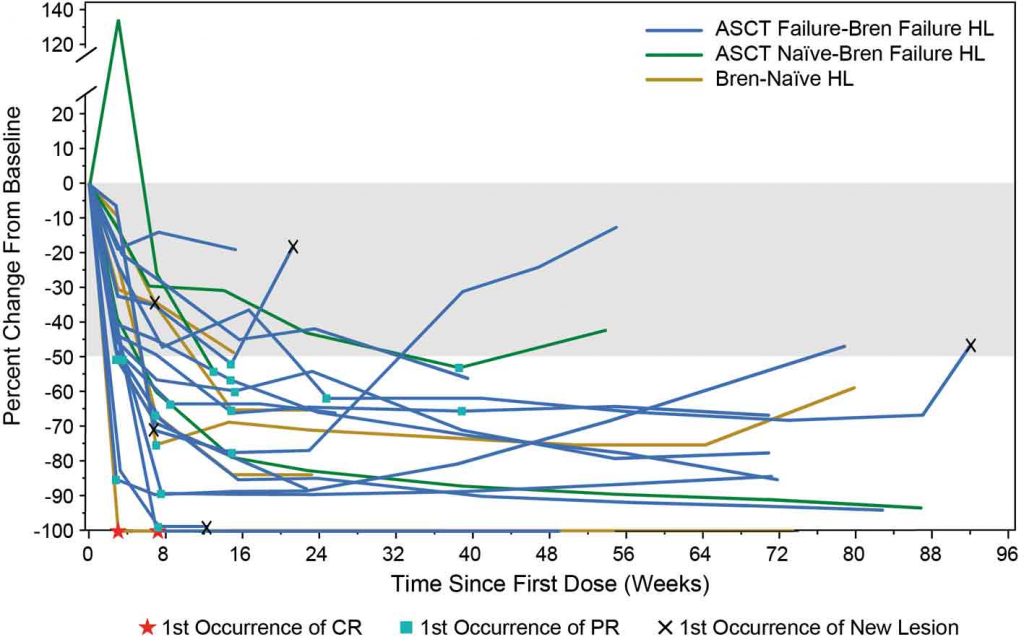
The phase I CA209–039 study showed that immunological response to nivolumab
in heavily pretreated patients with classical Hodgkin lymphoma is quite variable and individual for each patient
ASCT – allogeneic stem cell transplantation, HL – Hodgkin lymphoma, Bren – brentuximab vedotin Source: J Timmerman et al (2015) 13th International Conference on Malignant Lymphoma, Lugano, Abstract #010
Combination treatments including nivolumab
Many clinical trials have investigated the safety and efficacy of different combination treatments including nivolumab. A recent phase I/II trial investigated the combination of brentuximab vedotin with nivolumab in 42 patients with refractory or relapsed Hodgkin lymphoma, including 40% with primary refractory disease. Of these, 33% relapsed within one year and 26% relapsed after one year (Herrera et al. 2016, American Society of Hematology Abstract #1105). Treatment involved administration of cycle 1 of brentuximab given on day 1 and nivolumab on day 8, followed by administration of cycle 2 through 4 with brentuximab and nivolumab given in combination on day 1 and then every 21 days. After four cycles of treatment, patients’ responses were assessed, and those who were eligible could then choose to go on to ASCT.
Preliminary results showed a 90% objective response rate to treatment, with 62% of patients in complete remission (Deauville Score 1–3 on PET scan), 28% in partial remission, and one or two patients with stable or progressive disease. Almost two in five patients (38%) experienced infusion-related reactions to treatment, but the overall safety profile was manageable, with no dose reductions or discontinuations due to adverse events. The incidence of immune-related adverse events was low, and there was no antagonism between brentuximab and nivolumab. The promising activity supports further exploration of this chemotherapy-free regimen for relapsed/refractory Hodgkin lymphoma.
Looking to the future
There are now many clinical trials with different combinations of these new immunotherapy agents, used with other checkpoint inhibitors or other agents, both in first line and in the relapsed refractory setting. Immuno-oncology has changed the scenery for patients with relapsed/refractory Hodgkin lymphoma, with higher overall and complete response rates than traditional chemotherapy.
Nivolumab is currently licensed for patients with refractory or relapsed Hodgkin lymphoma, after ASCT and brentuximab. Toxicity is mostly immune related, but manageable with close monitoring.
Further studies are needed to explore the specific indication for these treatments and to evaluate the best combination and timing of treatment.
We also need to find a different way of assessing clinical response, with many patients showing a long-term partial response.
 In conclusion, it is very encouraging to have new options for this subgroup of Hodgkin lymphoma patients, and we are eager to gain further information on how best to use these drugs.
In conclusion, it is very encouraging to have new options for this subgroup of Hodgkin lymphoma patients, and we are eager to gain further information on how best to use these drugs.
Question and Answer session with Astrid Pavlovsky
Emmanuele Zucca, from the Oncology Institute of Southern Switzerland, Bellinzona, posed the questions.
EZ: I do not agree that brentuximab is chemotherapy-free and I would be more restrictive in the use of the term. I must also highlight that chemo-free does not mean side-effect free. Would you like to comment?
AP: Yes, I must highlight that we are dealing with something different, not necessarily less toxic, and we need more time to see what will happen in patients as they go along. Also, we must see what treatments we can use if immunotherapy were to fail, using information from clinical trials – including whether allogeneic stem cell transplant is an option – because there is limited experience of treatments after checkpoint inhibitors have failed.
EZ: In addition to brentuximab vedotin, do you consider any other agents worth testing in combination with nivolumab? Do you think we will see phase I trials exploring use of these treatments as first-line therapy, or do they have a role only in relapsed or refractory Hodgkin lymphoma?
AP: I think lots of patients do well with ABVD, so I think it’s unlikely that all patients will need these treatments as first-line therapy. They are also very expensive compared to traditional therapy. I think further evidence is needed from randomised trials to support the efficacy of these drugs as frontline therapy, and they must show greater clinical benefit than existing treatment [ABVD]. Many patients are cured with ABVD with good overall survival. These are very promising drugs, but we need more time and data to assess economic validity, toxicity and ease of delivery, to decide which patients receive these agents first line. For now, I think the role of these drugs is primarily for refractory and relapsed Hodgkin lymphoma.
With regard to combinations, there are many checkpoint inhibitors that have been combined with chemotherapy in clinical trials, including CTLA-4 inhibitors and PD-L1 inhibitors, and bendamustine has shown good results in refractory disease. Ipilimumab and pembrolizumab have shown promising response in this group of patients. We will soon have good data to support the best combination and to suggest which patients benefit most from particular combinations. However, it will be a challenge to analyse response to these agents in trials.