The debilitating impact neurotoxic drugs can have on patients long-term quality of life has been systematically underestimated. Can oncologists do more to pick up potential symptoms before they become irreversible?
Cancer patients about to start chemotherapy are often faced with a long list of possible side-effects, depending on the type of cancer and the drug or drug combination involved. They may also be facing side-effects from surgery and radiotherapy, so there is a lot to take in. Some side-effects, such as neutropenia, pose a serious risk, but only during treatment. Others, such as heart damage, may be irreversible, and have a lifelong impact on the quality of a patient’s life. Chemotherapy-induced peripheral neuropathy (CIPN) is one of these. Not only can it cause a range of mild to severe problems during treatment, but it can manifest itself strongly just after treatment, and can impact very heavily on a patient’s life for a long time, if not indefinitely.
CIPN is a condition that mainly damages the long nerves that extend to the feet and hands – a ‘stocking and gloves’ distribution – and can affect sensory or motor function, most often the former, with symptoms such as numbness and shooting pains. Some people are so severely affected that they may regret having the curative cancer treatment that caused it. Its mechanisms are not well understood, with little indication of who will suffer most, and there is no current treatment that can prevent it or do much to alleviate it. It affects 30–40% of patients across all neurotoxic drugs, but with some agents up to 70–90% of patients may be affected.
This presents a serious obstacle to successful treatment and, like all long-term effects, the problem is becoming more widespread as the population of cancer patients expands. While it only occurs in certain classes of chemotherapies used in some cancers, the treatments are among the more common, such as taxanes for breast cancer, and platinum agents for colorectal tumours (see table below).
Oncologists are often faced with a difficult decision about whether to stop or alter potentially life-saving treatment, and problems with neuropathy are a common reason for patients themselves deciding to stop therapy. Where severe problems arising from nerve damage do develop, health professionals also face the challenge of how to help patients to manage the symptoms.
CIPN has moved up the side-effects agenda in recent years, says Guido Cavaletti, a neurologist at Milan-Bicocca University, Italy, who has been treating and researching this condition for more than 20 years. “That’s because patients have been asking for a better quality of life as oncologists have been able to improve survival, and there has been a particular push for more attention to CIPN in the US, where patients tend to be more demanding, and as other side-effects can now be better managed,” he says. “But we still have virtually nothing for CIPN.”
Common symptoms
Cavaletti explains that the incidence of CIPN is much more frequent with the older chemotherapy drugs, although some of the newer biological therapies can also be neurotoxic. Numbness is the typical sensory symptom – being unable to feel heat or cold, or a pin prick. Motor symptoms include unsteadiness on one’s feet, or even being unable to walk far at all, in more severe cases. There are also pain symptoms: neuropathic pain is one of the most complicated pain types managed by neurologists or pain specialists, says Cavaletti.
What is a puzzle with CIPN is that, while it is known that nerve endings start to be damaged and can get progressively worse with chemotherapy, currently there is no way to tell what an individual’s risk is for developing neuropathy. “If I have ten patients, maybe two will develop severe neuropathy, four will have nothing and the rest some degree of symptoms – but we haven’t been able to identify the risk factors for who will get severe problems,” says Cavaletti.
Also unexplained is when CIPN will take hold, or what the symptoms will be. Dawn Storey, a consultant medical oncologist at the Beatson West of Scotland Cancer Centre in Glasgow, who has researched CIPN as part of her strong interest in supportive oncology, says: “The onset of acute neuropathy varies from patient to patient. Some get symptoms early on and have to stop treatment prematurely, whereas some get through treatment without many symptoms, but then may develop severe pain and disability after treatment stops – that’s what we call ‘coasting’, and it is often seen with platinum drugs.”
Although things do get better for the majority of people over several months, she adds, there are those who are left with long-term life-limiting symptoms, and little is understood about why patients develop different symptoms at different times, and why some and not others are left with long-term problems.
What is clear is the harm that can be done to patients, who can suffer a range of debilitating conditions. They may be relatively minor – Cavaletti mentions women who have had taxanes, who report they can no longer wear elegant shoes to the theatre as they feel too tight. “But imagine you are wearing boots and gloves permanently, or if you feel you have to wear gloves but it’s quite warm outside.”
If someone cannot feel the pedals in a car, they can’t drive, or they may be too unbalanced to walk far.
“If someone cannot feel the pedals in a car, they can’t drive, or they may be too unbalanced to walk far”
Storey says that oxaliplatin is a drug that tends to have different symptoms during the acute, treatment phase and post-treatment. In the acute stage, people can become hypersensitive to cold and experience difficulty swallowing – these usually go away in a few days, she notes. “Long term chronic symptoms tend to be the typical neuropathy, like numbness, shooting pains, pins and needles and problems with actions such as fastening buttons, writing and even toileting, as patients can’t feel the toilet paper.” Other drugs such as taxanes tend to have similar symptoms in both phases, although paclitaxel can cause a specific acute pain syndrome during treatment that affects the hips and trunk.
One group of cancer patients who have a particularly high incidence of CIPN are those with multiple myeloma. In this group, the nerve condition is not just a side-effect of the biological therapies, bortezomib (Velcade) and thalidomide, but can also develop as a complication to the myeloma itself. About 30% of patients are affected and it is a common topic of discussion at support groups. People report some awful symptoms of being unable to walk, pain and sleeplessness at night, and feelings of hopelessness.
Pain can be extreme to the extent that some patients can’t bear to have bedclothes over them, Storey adds. “I got interested in CIPN because I was disheartened that people were being cured of their colorectal cancer, but I was seeing them in their 50s or early 60s being unable to dress themselves, walk, write or drive safely, and losing their jobs because of their disability – that’s not success to me.”
As Cavaletti also says: “What we have now are cancer survivors living with severe damage. It’s a small group but we need to eliminate it. For example, there is a young psychologist colleague here who had testicular cancer two years ago and was cured with cisplatin – but now he cannot work as he can’t hold a pen. And he’s only 32 years old.”
An underestimated problem
Knowledge about CIPN’s incidence also appears to be lacking, although it is almost certain that long-term effects are underreported both in clinics and in trials. “Estimates of the proportion of patients affected by functional impairment due to neuropathy two years after the completion of treatment for colorectal cancer are about 4%, but it’s probably much higher than that,” says Storey. “In audit work I have done it seems that about 20–30% have some sort of impairment – and the disparity arises because published studies focused more on the anti-cancer effect of the chemotherapy rather than reporting long-term side-effects, which some oncologists rate as less important.”
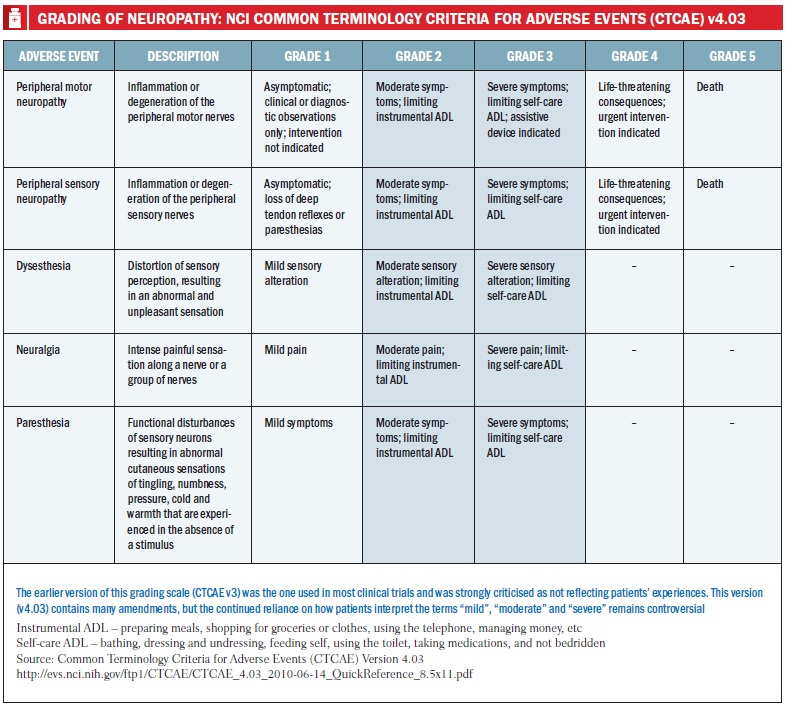
Another contributing factor is partly a result of the way that CIPN is assessed, she adds. Long-term studies used in guidelines have adopted the US NCI’s Common Terminology Criteria for Adverse Events (CTCAEv3) – one of several tools that can be used for CIPN – and most often significant neurotoxicities have been reported only at grade 3, which is ‘severe interference with daily living’. But distinguishing this from grade 2 – ‘moderate’ limits on daily living – can be an artificial judgement, says Storey, so “CTCAEv3 is now recognised as unfit for purpose in assessing CIPN. CTCAE v4 is a slight improvement, but I would still encourage investigators to report grade 2 and 3 because functional impairment of any degree matters hugely to patients.”
In practice, this has probably resulted in considerable harm. “From the point of view of most oncologists, if it’s grade 3 the drug is stopped, but if grade 2 it tends to continue to be given – so oncologists have probably been giving far too much oxaliplatin and caused more nerve damage than was realised. There is no official guidance on applying the criteria and oncologists tend to be biased in favour of giving a drug rather than stopping it, because of course they want to prolong life or prevent a cancer from coming back.”
A recent indication of the extent of long-term CIPN in people given oxaliplatin for colorectal cancer was reported at the EORTC survivorship summit by Lonneke van de Poll-Franse, from Tilburg University in the Netherlands. A quality of life questionnaire was sent to more than 1600 patients, on average six years after diagnosis, 500 of whom had received chemotherapy. About one in three of those who had been treated with oxaliplatin reported tingling, painful hands or feet up to ten years after diagnosis, and indicated that this CIPN “tremendously impacted on quality of life” across scores for social and physical functioning, and overall health.
Oncologists who specialise in cancers such as colorectal and multiple myeloma will tend to be more experienced with CIPN, as they see it most often, and they may therefore stop or reduce doses and switch to alternatives earlier. But there needs to be both a refinement of the assessment tools and greater interest in neuropathy among oncologists, according to Cavaletti.
While there are the usual centres of excellence in cancer – Storey for example did her work on CIPN at the Edinburgh Cancer Research Centre, where there are palliative and supportive care specialisms – Cavaletti says that up until the last few years it was common in some countries for neurologists to carry out much of the research on CIPN, and indeed to see patients, when oncologists should really have been doing more.
“Oncologists should be prepared to properly recognise and score the severity of CIPN and not just send patients to us once they are sure there is neuropathy – that’s not very useful for the patient.” Cavaletti makes a comparison with neuropathy caused by diabetes. “I don’t see these patients unless they are very different from usual – they are well managed by diabetes specialists, and we need to achieve the same with oncologists, some of whom are wary of a neurological exam – they think it is complicated and difficult but that’s not true.”
The critical point in managing CIPN during treatment is not to wait until it’s too late: the treatment should be changed before symptoms become so severe that they are irreversible. In the absence of effective treatment for CIPN itself, this is about the only primary strategy of value, says Cavaletti. He agrees that the NCI’s CTC scale is inadequate and a more robust method is needed to give oncologists the confidence that, for example, mild symptoms are not progressing so there is no need to move to a lower or less frequent dose, or another agent, or discontinue chemotherapy.
“It’s about managing the patient and not the side-effect that emerges,” he says, noting that when thalidomide first became available for trials in multiple myeloma, there were concerns that it would need specialist neurologic monitoring. “Now, in work on producing an improved assessment tool, it is clear that in 99% of cases a clinical evaluation is sufficient – you don’t need instrumental evaluation.”
But that clinical evaluation must comprise two elements, he adds – both the doctor’s assessment of nerve changes and the patient’s own report, although initially it can seem that the different viewpoints can be hard to reconcile. “But it is the same problem from two perspectives, and is particularly important as we have no objective tools to say measure pain in a patient. We don’t have an operational patient-reported outcome measure in CIPN yet, but we are working on it and combining it with clinical evaluation is a goal for our research.”
Asking patients about symptoms, and ensuring they understand the potential long-term implications, is particularly important, as studies have shown that patients often don’t like to mention side-effects for fear that their treatment might be stopped.
As Storey adds: “We also know from research that patients are more likely to disclose their symptoms to a nurse than to a doctor, who patients often assume is more focused on cancer management than on issues such as fatigue, anxiety, depression and CIPN. And, patients who are cured often don’t want to sound ungrateful, saying they can put up with CIPN – but I know some who have said they would not have taken the chemotherapy if they had known it was going to make them feel that bad. But most won’t tell you and they certainly won’t if you don’t ask.”
 Improving evaluation
Improving evaluation
Cavaletti is principal investigator for the CI-PeriNomS group, which is testing existing scales for assessing CIPN together with quality of life tools, including the EORTC’s QLQ (quality of life questionnaire), with the aim of producing a standardised outcome measure, and a new cohort of patients is being assessed this year. “We had our first meeting in 2007 and while we have expanded the study group to 20 centres, most in Europe and some in the US, we have been unable to get any support for our work,” he says. Most of the researchers are neurologists, but more oncologists have come on board, he adds.
Storey, who has been involved with the group, feels part of the answer lies in better communication with patients, including at the start of treatment. “I discuss drug options at length with patients, asking about hobbies, such as knitting or playing an instrument, that CIPN could adversely affect.” The point is echoed by van de Poll-Franse, who was involved in the study of patients treated for colorectal cancer. She agrees that quality of life should be part of the discussion when considering oxaliplatin as an adjuvant therapy.
“Part of the answer lies in improved communication with patients, including at the start of treatment”
There are various treatments that can alleviate symptoms in some people, and an increasing number of trials are investigating which work best for CIPN. Antioxidants, antidepressants, anticonvulsants (such as pregabalin), opioids and analgesics, including topical substances such as menthol, and acupuncture, are all in the frame. Storey has trialled menthol cream and found a good response to pain, although she says it may worsen it in some cases.
Newly published guidelines from ASCO recommend treatment with the antidepressant duloxetine, and suggest that gabapentin, and a compounded topical gel containing baclofen, amitriptyline HCL, and ketamine may also be used, as their utility has been shown in other neuropathic pain conditions.
Finding biomarkers and genomic information that can predict who will suffer most is also a research avenue, and of course more personalised treatment will cut the number of patients receiving neurotoxic drugs in the first place.
Genetic information may indeed be the best path, as Charles Loprinzi, a medical oncologist at the Mayo Clinic in Rochester, US, and an expert in CIPN, has recently noted. There were hopes for a prevention approach for oxaliplatin by infusing calcium and magnesium, but a recent trial he led has proved negative (JCO doi:10.1200/JCO.2013.52.0536).
The NCI guidelines are unable to offer any recommendation on prevention, due to the lack of high-quality consistent evidence.
As Cavaletti laments, there is much more interest in funding work on CIPN in the US, where he says that the National Institutes of Health (NIH) has launched a programme to discover the mechanisms that cause it, and there have also been meetings on improving outcome measures in clinical trials, which have had strong participation from patient associations. “To my knowledge there is nothing like this in Europe and it’s a gap we need to fill,” he says.
Storey agrees, saying that research funding in palliative and supportive care has been curtailed in the UK, and that not all cancer centres have the multidisciplinary resources of Edinburgh, where there is access to neurologists, anaesthetists and palliative care colleagues for problematic cases. In fact she says that some of the most useful help that her patients receive is from colleagues in occupational health. In a pilot in Glasgow they are helping with adaptations in the home, such as foam handles for kettles, elastic shoelaces, temperature checks for bathwater. “We must talk to patients about CIPN because they are often battling on their own,” she says.