Many patients are being treated with costly new radiotherapy treatments on the basis of hype rather than evidence that they stand to benefit. For others, lack of evidence is delaying access to new treatments that could make a real difference. Janet Fricker reports on efforts to develop a scale to measure the value of locoregional cancer therapies, and asks: could it help us make better use of high-tech interventions?
Over the past few years an impressive array of new technology has become available in radiation oncology. Together with improved diagnostic imaging and better computer algorithms, advanced radiotherapy technologies have evolved treatments from using simple rectangular radiotherapy fields towards techniques such as intensity modulation and stereotactic targeting that focus the beams more precisely on the tumour.
In parallel, the nature of the beams themselves has evolved. In addition to the traditional photon beams delivered by linear accelerators, cyclotrons are now able to deliver beams of much heavier protons and carbon ions. Other innovations include shorter fractionation schedules, motion management and adaptive radiotherapy, novel combinations with systemic drugs, superior image guidance (using MRI as opposed to CT for greater soft tissue resolution), and new immobilisation systems.

“The overall result is that we’re better able than ever before to target tumours and spare the surrounding critical organs from toxicity,” says Yolande Lievens, chair of the radiation oncology department at Ghent University Hospital, Belgium. But as she acknowledges, none of this high-tech equipment comes cheap, and nor does the additional expertise needed to carry out the imaging, planning and delivery of each treatment.
Convincing health services to introduce these new treatment modalities into everyday clinical practice will require demonstrating that the benefit they deliver is worth the additional cost.
“With so many developments, we need to start to judge value in radiation oncology and define what works best in different clinical scenarios. We want to ensure we aren’t using a sledge hammer to crack a walnut and that all these new high-cost technologies deliver real clinical benefits,” she says.
Lievens is now leading efforts to develop a framework for assessing the value of radiotherapy and surgical – i.e. ‘locoregional’ – procedures, in much the same way as ESMO’s Magnitude of Clinical Benefit Scale, and its US equivalents (the ASCO Value Framework and the NCCN Evidence Blocks) do for systemic therapies.
A new (as yet unnamed) European group has been set up under the auspices of the European Cancer Organisation (ECCO) umbrella, with Lievens in the chair, tasked with developing a value framework that could be applied across radiation oncology and surgical techniques and across different treatment settings.
If we want to provide the best care…
The rationale for developing such a yardstick is widely accepted, and goes well beyond radiation oncology, as Lievens explains. “Across Europe cancer expenditure is rising exponentially, driven by the growing ageing population, numerous therapeutic advances and expanding choice and consumerism in healthcare.”
Cancer care costs health services more than any other disease, with the American Institute of Cancer Research estimating that in 2016 the world budget for oncology treatments was $895 billion. For sustainable health systems, there is growing recognition of the urgent need to define the patient groups who need the most advanced treatment approaches, as compared with those who would do just as well with standard cheaper treatments.
In a position paper calling for public policy debate on access to cancer innovations, Matti Aapro, President-Elect of ECCO, argued that ‘newer’ may not necessarily equate to ‘better’, and that ‘older’ alternatives, and ‘simple’ interventions may deliver the greatest impact on improving patient care (EJC 2017, 82:193–202).
“We need to apply more scrutiny to the way we deliver care today, be ready to remove or discontinue practices or interventions that are inefficient, and be forward-thinking to prioritise innovations that may deliver the best outcomes possible for patients with the resources at hand.”
His point is echoed by Ajay Aggarwal, a clinical oncologist specialising in prostate cancer at London’s Guy’s and St Thomas’ Hospital, who is working with Lievens on the value framework. “Access just because a treatment is new is not something we should be striving for. We need to have shown clearly that access to new treatments will bring meaningful improvements in the quality and length of life, and can reduce the toxicity and financial burden to patients associated with treatment.”
Adapting the medical oncology benefit scales
The ESMO Magnitude of Clinical Benefit Scale (ESMO-MCBS) was developed to provide a ‘rational, structured and consistent’ approach for ranking relative benefits of drug treatments in solid tumours, to help with priority setting and decision making in the face of large numbers of new and costly cancer treatments coming onto the market.
As Elisabeth de Vries, chair of the ESMO-MCBS working group, explains, that exercise too was about delivering the best possible outcomes with limited resources: “In cancer there’s a profound problem with numerous new drug treatments, but only finite amounts of money, making it necessary to discriminate drugs that really make a difference to patients,” she says.
Advances in radiation oncology
Traditionally, external beam radiation therapy (EBRT) uses a linear accelerator (LINAC) to deliver photons (high-energy X-rays) to tumours. More recent advances include improved image guidance and computer algorithms, allowing radiotherapy to evolve from simple treatment fields towards highly conformal radiotherapy techniques, such as intensity-modulated radiotherapy (IMRT), and stereotactic radiotherapy (SRT).
- IMRT enables dose to be shaped to tumours by modulating (controlling) the intensity of the radiation beam, allowing different doses of radiation to be given across the target. CT scans map tumours in 3D, with computers controlling machines fitted with a multi-leaf
collimator, composed of thin lead leaves, to shape radiation beams precisely to tumours.- Stereotactic radiotherapy (SRT) uses 3D coordinate systems and/or advanced imaging to locate small tumour targets. It was originally delivered with brands including CyberKnife and Gamma Knife, but it is now most frequently delivered with standard LINACs. It was used first for brain and spinal cord tumours, but development of stereotactic body radiation therapy (SBRT) allowed the principle to be transferred to other indications such as lung, liver, pancreas and prostate cancers.
Such conformal approaches have improved outcomes by escalating dose to targets and minimising toxicity to normal tissue and critical organs. Treatments can be delivered in one to five treatment sessions compared to typically five to eight weeks for standard external beam radiation therapy. More detail on these advances can be found in a review by SS Ahmad and colleagues (BMJ 2012, 345:e7765).
Conformal radiotherapy techniques can be adapted to deliver heavier particles such as protons and carbon ions, produced by particle accelerators (cyclotrons), as alternatives to photons.
- Proton beams (charged nuclei of hydrogen atoms) have a peak of dose deposition at a sharply defined point (the ‘Bragg peak’) allowing for a much lower dose to nearby critical organs. Proton therapy is most widely used for treating tumours located close to vital organs that would be unacceptably damaged by X-rays, or in paediatric oncology where late side effects are of major concern. According to the Particle Therapy Co-Operative Group, around 80 proton beam facilities exist worldwide, with around 30 now operating in Europe.
- Carbon-ion radiotherapy (CIRT) uses charged carbon nuclei particles, which have a larger mass and greater charge than protons, yielding even sharper dose distributions. Another advantage is that its efficacy is unaffected by the low oxygen levels occurring in tumours, so it is more effective in radioresistent tumours. Although CIRT has been used since 1994 to treat cancer in Japan, it has only recently been available in Europe, with facilities now operating in Vienna, Heidelberg, Marburg, and Pavia. The lack of centres has made phase III trials difficult. Photons, protons, and carbon ions all kill cells in different ways, making it important to establish the different tumour types where each is most effective.
Under the MCBS system, systemic therapies are judged on a range of factors including overall survival, progression free survival, hazard ratios, long-term survival, response rates, prognosis, quality of life and toxicity. For curative settings, the therapies are graded A, B, or C, with grades A and B representing a substantial level of clinical benefit; while for the non-curative setting, the scale is graded 5,4,3,2,1, with grades 5 and 4 representing substantial levels of clinical benefit.
The question being asked by Lievens and her colleagues is: how can this scale be ‘nuanced’ to reflect the specificities of locoregional treatments – both in radiation and surgical oncology. Their initial conclusions, published earlier this year (Lancet Oncol 2019, 20:e112–23), indicate that such an exercise could be possible – but will take a lot of time and effort.
End points
One of the first differences the review authors identified between systemic and radiation oncology treatments was relevant trial endpoints. Aggarwal, a co-author of the paper, explains that, while both approaches consider overall survival, in radiotherapy far greater emphasis is placed on local control, organ preservation and acute and late toxicity, which are not weighted in the current value frameworks. Progression free survival is considered of far less importance in this setting.
“Local control represents organ function aspects like the ability to swallow in oesophageal cancer or control urination in bladder cancer. These are the outcomes which really affect patient wellbeing,” says Aggarwal. He also points to the need to take into consideration the late clinical effects on organs beyond the cancer, such as rectal bleeding and chronic diarrhoea, that can result after radiation oncology for prostate cancer.
A clear distinction in radiation oncology trials needs to be made between acute toxic effects (that occur within three months of treatment), and late (chronic) toxic effects (that occur months or even years after treatment). “With more patients cured with radiotherapy than systemic treatments, the issue of survivorship quality becomes much more pertinent,” he points out.
Progress in technology or outcomes?
For radiotherapy trials, it will be important “to focus on innovations that represent real changes in the treatment process that could advance our ability to control the cancer or reduce toxicity,” such as new fractionation schedules or the addition of systemic drugs to standard radiotherapy, stresses Aggarwal.
He contrasts these with “incremental innovations that are likely to achieve similar outcomes to established technology, but to do so more efficiently.”
Types of evidence
Different levels of evidence beyond formal randomised controlled trials might be considered for radiation oncology, they suggest, such as model-based studies and real world evidence.
Cai Grau, a radiation oncologist at Denmark’s Aarhus University, who has done a lot of health economics work with the European radiation oncology society ESTRO, is another co-author of the study. He explains that modelling – exploring how therapy doses affect patients according to their individual anatomy – can be performed to predict benefits without the need for randomised trials. The approach can also be used to ‘enrich’ trial populations, he says. “The principle is more or less the same as testing for biomarkers before enrolling patients in targeted therapy trials. Modelling allows you to perform studies in specific cohorts of patients where you know there is likely to be benefit.”
The study also suggests that registries can be used to explore the intervention in real world populations, allowing consideration of patients who do not meet the stringent criteria of clinical trials. Aggarwal takes pains to caution, however, that the quality of the data is only as good as the registry infrastructure.
Greater emphasis is placed on local control and organ preservation, which are not weighted in current value frameworks
“Registries in radiation oncology need to have near complete coverage of the relevant population, with low levels of missing data, and ensure that endpoints in addition to survival can be captured, such as adverse events from treatment as well as markers of disease progression or relapse.” This needs considerable methodological input and time, stresses Aggarwal, who challenges the “often heard assumption that any available patient-level real world data is relevant and can be used to inform practices of care.”
“Using a combination of modelling and real life data you can get a feedback loop to define the types of clinical questions that you want to pose in a trial,” says Aggarwal.
Then there is the issue of accounting for the expertise of the provider. Radiation oncology is more dependent on healthcare provider expertise than systemic treatments, says Lievens. “Consequently, any benchmarking of value will need to take into consideration the quality of delivery.”
The patient perspective
New tools will be needed to identify the aspects of care that matter to patients, such as shorter and less invasive treatment schedules, and the ability to return quickly to normality and work. This may be trickier than it looks. As the review acknowledges, patient values are not a ‘one size fits all’, but are influenced by external factors such as social, religious and cultural environments as well as patient-specific factors, such as gender, education, and personal finances.
“It’s important for scales to distinguish living longer with better quality of life from living longer with worse quality of life,” says Bettina Ryll, founder of the Melanoma Patient Network Europe, who has been providing patient input for the ESMO-MCBS scale. “There is a need to measure what matters to patients and this varies between individuals. For example, not being able to walk far would have an entirely different dimension for a marathon runner compared to a couch potato.” Tools, she adds, should be able to take into consideration potential trade-offs, for instance between short-term severe toxicity and low-grade but long-term toxicity.
“Trials need to focus on innovations that represent real changes that could advance our ability to control the cancer or reduce toxicity”
Values are not static, cautions Kathy Oliver, founding director and chair of the International Brain Tumour Alliance, who contributed the patient advocacy perspective to the Lancet Oncology study. “They alter as the patient’s journey unfolds and he or she travels through illness, treatment, survivorship, and potentially end of life. During some of these stages, non-interventional support may be of huge value to patients, this includes access to patient organisations, support groups, clinical nurse specialists, rehabilitation and palliative care. But these types of support are rarely acknowledged as being of importance in value frameworks.” Added to this, says Oliver, cancers can be very different from each other, with varying side effects, symptoms and outcomes. “Short prognosis conditions may call for different value scales than longer-term conditions, taking into account trade-offs between benefit and risk and extended survival versus quality of life.”
Value for money
The group believe that it will be important to include economic endpoints to define more explicitly the financial costs of new innovations. This is something the ESMO-MCBS was careful not to do, partly because costs vary so much across Europe.
de Vries, who chairs its working group, says excluding cost considerations also “gives freedom to think what the scale really means for patients”. She adds, however, that ESMO does now see the value of addressing financial aspects, and is exploring whether it might be possible to incorporate the ESMO-MCBS in a geographically based reimbursement model.
Ending hype-driven decision making
The hope is that having a single scale by which to judge the value of new technologies used in locoregional cancer treatments will help ensure decisions on investment and deployment of new technologies are taken on the basis of evidence not hype.
Aggarwal points to the experience with the introduction of DaVinci robotic surgery systems as an example of the latter. When in 2006 the UK’s National Health Service allowed greater choice over where patients received treatment, many men with prostate cancer opted to attend robotic centres. Resulting market forces led to a rise in the number of centres offering robotic surgery from 18% (12 centres) at the beginning of 2010 to 71% (39 centres) at the end of 2014 (Lancet Oncol 2017, 18: 1445–53), with nearly 90% of all centres offering robotic surgery for prostate cancer.
“The growth was despite a scarcity of evidence for superiority of robotic surgery with respect to both functional and oncological outcomes, and the procedure costing far more than conventional open surgery,” says Aggarwal. “It’s human nature to assume that the latest innovations are better, and should replace older more established treatments,” he adds – which is exactly why robust quality performance measures, are so badly needed.
In the United States, such ‘human assumptions’ have been used to market proton therapy for a variety of cancer indications. Men with prostate cancer have been a target, because they are a large market, and because one of the ‘unique selling points’ of proton therapy is that protons deliver most of their dose at a particular point rather than along their entire beam trajectory, which offers the potential for protecting organs on the far side of the target.
However, unlike the many other options for treating prostate cancer, the evidence base for proton therapy is small and conflicting. One of the few studies comparing conventional with proton beam therapy for prostate
cancer found that gastrointestinal problems were in fact worse in the group receiving proton beams (Eur Urol 2011, 60:908–16). In addition, many men who opted for proton treatment would not have required any treatment at all beyond ‘watchful waiting’, because their disease was unlikely to progress in a clinically meaningful way during their lifetimes.
And this uncertain benefit does not come cheap. A new proton beam therapy service at The Christie Cancer Centre in Manchester, UK, cost around €145 million to develop. Little wonder then that, in the absence of any restraints, US healthcare providers who invested in this technology were driven to aggressive marketing to pay off the loans.
The impact of ESMO’s Magnitude of Clinical Benefit Scale
Since 2015 The European Society for Medical Oncology has applied its Magnitude of Cinical Benefit Scale (ESMO-MCBS) prospectively to new anti-cancer interventions approved by the European Medicines Agency (EMA), with ESMO guidelines incorporating scale results. It is also used by the World Health Organization to support selection of cancer drugs for their essential medicines list and by patient advocates to lobby for access to drugs that make a real difference. The scale is now starting to be incorporated into health technology assessment processes. Notably, one middle-income country experiencing huge problems with drug prices used the ESMO scale to define their essential medicines list, with the result that they were able to maintain universal health coverage. “Ultimately, the scale helps doctors and patients to sit together to discuss whether they want to use a drug or not,” explains Elisabeth de Vries, who chairs the ESMO-MCBS working group.
Richard Sullivan, a member of the initial ESMO-MCBS task force, believes the scale provides a vehicle to slice through the hype of clinical trials, distinguishing cancer therapies with trivial clinical benefits delivering progression free survival advantages of a few weeks from drugs that can substantially improve long term survival. “Just because a drug has received marketing authorisation, the trial has been published in a reputable journal, and the press release proclaims a statistically significant result doesn’t mean that the drug is of real value,” says Sullivan, from Kings College, London. “The scale provides a mirror allowing you to take a hard-nosed look about whether reported outcomes are clinically meaningful.”
For Cai Grau, however, the inappropriate use of expensive high-tech treatments is only one side of the problem. Perhaps a greater concern for him is that public health services, where academic research is located, have been reluctant to invest in such expensive technologies when the commercial sector already has more than enough capacity to treat the limited number of patients for which there is evidence of benefit. But the commercial providers, who now carry out the lion’s share of proton therapy procedures, have minimal interest in performing the trials that are so badly needed to generate evidence on whether new technologies like this really do deliver better outcomes, and for which patients and indications.
Towards a culture of value-based research
The problem, says Grau, is not limited to proton therapy. “Unlike drugs, where the vast majority of trials are undertaken by pharma, in radiation oncology it is largely left to the professional community of independent investigators to assemble the evidence,” he says. A key reason is that manufacturers only have to demonstrate their devices are safe to use, but not the impact on outcomes. Research in radiation oncology is therefore heavily dependent on public funding, which is becoming harder to come by as the technologies become more sophisticated and costly.
“We’ve experienced something of a catch 22 situation in radiation oncology, where many countries want evidence that therapies work in different cancer locations before investing money in infrastructure. But the reality is that you can’t undertake research until you have invested in equipment.”
Aggarwal agrees that much more needs to be invested in generating evidence in radiation oncology. Given that this type of treatment contributes to 40% of all cancer cures (Nat Rev Cancer 2009, 9:134-42), yet accounts for just 5% of the overall cancer treatment budget (Acta Oncol 2003; 42: 357–65), it makes sense to invest more in maximising the value for patients.
Aggarwal, Grau and Lievens all hope that developing a single, evidence-based and consistent approach to measuring that value will help win the argument with funders about the value of investing in that research. The prize will be moving towards health systems that promote innovation, avoiding delays in clinical adoption of valuable new treatments and preventing widespread adoption of interventions that offer no benefit or can result in harm.
The Lancet Oncology paper is just the beginning of the beginning. It makes a strong case for both the urgent need and the feasibility of developing such a value scale for locoregional treatments. The authors are under no illusions, however, about the length of the journey they plan to embark on. “We’re well aware that it will be a really labour-intensive effort that will need additional funding and support,” says Lievens.
The group are also mindful that cancer is treated by multidisciplinary teams, and of the need to undertake a whole-system approach to innovation across systemic and locoregional interventions. “Ultimately, we’ll need to find a way of aligning value scales for systemic, surgical and radiation oncology so that we can capture in a reliable way what’s best for the individual patient,” she says.
Proton beam research: catching up with clinical practice
Europe has generally been slow to invest in proton therapy facilities. However, the number of academic centres actively delivering proton therapy in the EU is progressively expanding, with established centres in Germany, Belgium, Switzerland, Sweden, France, Italy and the Netherlands. The launch of The Christie NHS Foundation Trust Proton Beam Centre in Manchester, UK, in autumn 2018, and the Danish Centre for Particle Therapy in January 2019, together with the anticipated launch of the University College London Hospitals Proton Therapy Centre in the summer of 2020, will further boost Europe’s capacity to carry out collaborative research on this unique form of radiotherapy.
Research activities will be in addition to providing a routine proton service for established clinical proton indications. These are currently limited largely to highly complex brain cancers, head and neck cancers and sarcomas, particularly in children, where reducing radiation doses to normal tissue avoids adverse effects on growth, intellectual development, endocrine function, and secondary cancer development. The evidence-based guidelines originated from the American Society for Radiation Oncology (ASTRO) after observational studies at Loma Linda University and the Massachusetts General Hospital.
This new investment comes almost a decade after proton therapy took off in the US market with aggressive marketing campaigns around many indications for which the theoretical advantages of protons have not been confirmed in randomised trials, including prostate, liver, pancreas and lung cancers. The failure to generate strong evidence on the value of proton therapy in these cancers is because proton beam therapy has been mainly performed in privately funded centres that do not undertake research, but seek to maximise their market to make a profit on the huge upfront investment required for proton therapy facilities.
In Europe, the establishment of government-funded proton treatment centres in recent years has resulted from strategic national health business plans estimating that it is more cost effective for health services to set up their own proton centres.
One of the important spin-offs from increasing the number of government funded proton centres across Europe is that ‘protected beam time’ will enable both basic science and clinical research, says Neil Burnet, from the University of Manchester.
“Together with established centres in Holland, Switzerland, Germany and Austria, we’re reaching a critical mass of proton facilities where we are starting to have enough centres to undertake international collaborative research efforts,” he says.
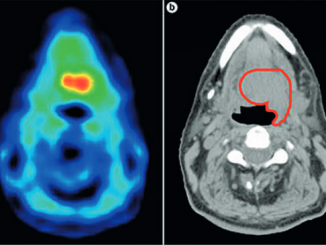
One of the first trials planned for patient populations that do not meet the current ASTRO proton therapy guidelines is the phase III TORPEdO trial at The Christie, where intensity-modulated proton therapy is being compared with intensity-modulated radiotherapy in oropharyngeal cancer. “The key thing will be to explore whether protons reduce treatment side-effects, such as difficulty swallowing and dry mouth, in a way that’s useful to patients,” says Burnet.
Other trials in similar populations of patients are planned in the Netherlands and Denmark in the hope of eventually performing a meta-analysis. No less important, says Burnet, will be the opportunities the new facilities offer to undertake physics-related research exploring where proton beams stop, and cell culture work exploring synergies between protons and systemic treatment.