If we can only find out what is driving a given cancer and/or what mechanism is creating resistance to treatment, it should be possible to at least halt the progress of the tumour by blocking the driver or the cause of the resistance.
Cancers – particularly advanced solid tumours – are much too complex to be controlled through such precisely targeted treatments. They are too heterogeneous, and their capacity to mutate enables them to find a way around any roadblocks we can throw at them.
 This argument has been going on for at least 50 years, and was brought into focus in 1984 with the classic Cancer Research paper by Gloria Heppner on tumour heterogeneity and its impact on therapy. Proof of principle that the personalised approach can work came in the late 1990s with trastuzumab, the first anti-cancer drug designed to block the mutations driving HER2+ breast cancers, followed by imatinib, which proved highly effective at controlling chronic myelocytic leukaemia – a blood cancer driven by a single mutation. But hopes that targeted therapies can transform advanced solid tumours into manageable chronic diseases are still a long way from being realised.
This argument has been going on for at least 50 years, and was brought into focus in 1984 with the classic Cancer Research paper by Gloria Heppner on tumour heterogeneity and its impact on therapy. Proof of principle that the personalised approach can work came in the late 1990s with trastuzumab, the first anti-cancer drug designed to block the mutations driving HER2+ breast cancers, followed by imatinib, which proved highly effective at controlling chronic myelocytic leukaemia – a blood cancer driven by a single mutation. But hopes that targeted therapies can transform advanced solid tumours into manageable chronic diseases are still a long way from being realised.
Is it just a case of giving this approach more time to define more mutations and develop more drugs to block them? Or should we be asking questions about whether we may be betting too heavily on the personalised cancer medicine paradigm, and whether we should redirect some of the research effort into areas that are more likely to deliver the big differences that everyone wants to see?
John Hickman, now an independent consultant who worked as a researcher in academia, and more recently in the pharmaceutical industry, believes we should be asking questions. In an article published in the New England Journal of Medicine, Hickman and his co-author questioned what evidence there is to show that the personalised cancer medicine paradigm will ever deliver big benefits to large numbers of patients, and called for a proper evaluation of the strategy “in a small number of well-designed collaborative programs” (NEJM 2016, 375:1289–94).
Giuseppe Curigliano, Professor of Medical Oncology at the University of Milan, and Head of the Division of Early Drug Development at the European Institute of Oncology, Milan, says that is never going to happen. He argues that new targeted medicines are proving their value in patients every day, and that treating cancers according to their molecular profile is the future.
Cancer World invited Curigliano and Hickman to debate the topic to help clarify the key differences between their two perspectives, and see if they could find any common ground.
*******
 John Hickman
John Hickman
“Our cancer research efforts are currently invested very heavily in personalised cancer medicine, a paradigm that is based on the concept that molecular analysis of a metastatic tumour in an individual patient will allow the selection of effective drugs to control that tumour and thereby significantly prolong survival of the majority of patients undergoing this treatment. This paradigm is appealing to patients and to foundations that support cancer research, but there are strong grounds for questioning what it can deliver for the large majority of patients with advanced solid tumours either now or in the future. The cancer community has a duty to ask those questions, which is what I did with Ian Tannock in writing our New England Journal of Medicine article on the Limits of Personalized Cancer Medicine (NEJM 2016, 375:1289–94).
The essential question for personalised cancer medicine is whether any therapeutic strategy could provide cure or long-term remission despite the presence of inter- and intra-tumour heterogeneity, the heterogeneity of tumour microenvironments and the almost universal capacity of cancer cells to develop resistance, reflecting the enormous plasticity of tumour biochemistry. Two decades on from the approval of the first HER2 inhibitor trastuzumab, that question has yet to be answered.
There are high costs associated with focusing research and development resources so heavily on pursuing the personalised cancer medicine paradigm. The individual drugs are expensive, often with marginal benefit, and they are increasingly used in combinations, escalating both financial cost and toxicity. More seriously perhaps is the cost of continuing to plough resources into a strategy that has yet to show any benefit, rather than spreading the research effort across a wider range of promising avenues such as the early detection and treatment of major cancers.
To clarify the likelihood that personalised cancer medicine will be able to deliver on its initial promise, what is needed is to see a clear impact on patient survival and quality of life together with estimates of cost–benefit. This should come from a small number of well-designed collaborative research programmes. The cancer research community has a responsibility to ensure these are urgently undertaken, and transparently reported on, to ensure time and resources are not wasted pursuing a questionable strategy”.
 Giuseppe Curigliano
Giuseppe Curigliano
“I disagree. All the achievements from the past 10 years have derived from precision medicine. ALK-amplified non-small-cell lung cancer is a good example. It is by sequencing that we discovered that target and then we developed a new generation of ALK inhibitors. The same applies to trastuzumab, gefitinib, erlotinib. It is a matter of discovery and drug development.
I also disagree completely that there is a minimal impact on survival and quality of life. If you discover an ALK amplification in a lung adenocarcinoma and you give an ALK inhibitor, you will increase the survival of the patient. And since the patient will not receive chemotherapy, you will increase also their quality of life. Before ALK inhibitors, survival was three months, and now it is three years. It’s the same for BRAF-mutated melanoma. We already have in real life an achievement that completely changed the landscape of treatment over the past five years.
What we are doing now is to discover the mechanisms of resistance to these agents and to develop new agents that are more effective. The landscape from now to 10 years’ time will see increased activity of targeted agents – which in my opinion will lead to improved outcomes in terms of overall and progression free survival as well as quality of life, because the new agents are less toxic.
In the future, our sequencing technology will permit a deeper analysis of genetic changes. We will discover new genetic alterations, and should look at the patient population in the future not as a general population but as niches of populations. So you will have 5% with a PI3K mutation 2% with a BRAF alteration, 5% of patients with an HER2 mutation. Some subgroups of patients will benefit from one approach, some from another. From the sum of all the subpopulations we will capture the overall population. In this overall population you will have a single patient with his or her individual mutation profile, permitting personalised treatment
Every day there is a new mutation, a new alteration, a new biomarker being identified. We are moving from disease-oriented treatment to pathway-oriented treatment. Just think about pembrolizumab, the first cancer treatment to be approved by the FDA not for a specific cancer, but for any advanced solid tumour with flaws in genes that repair DNA. More are expected soon.
In the last 10 years, we have approved many more molecular entities in terms of new treatments than in the last 30 years”.
 John Hickman
John Hickman
“I agree that the 3 to 5% of patients with lung adenocarcinoma carrying an ALK translocation do indeed have a prolonged survival and it is important that patients who will benefit get access to ALK inhibitors. My concern is that there are a priori reasons to question whether the ‘ALK paradigm’ will work for the majority of patients, or whether it will even deliver to ALK+ patients the long-term control or cure that they need.
I question an approach where large numbers of patients with advanced, metastatic cancers are selected for treatment with targeted agents (‘basket trials’) based on the presumption that a biopsy (often only a single biopsy), and genomic sequencing, will indicate that tumour’s drug sensitivity. Randomised trials of this approach have yet to validate its impact on survival for the large numbers of patients involved, or to measure its cost-effectiveness. The SHIVA trial was the first reported ‘basket’ trial of this type, which compared standard of care against treating a variety of solid tumours based purely on their genomic profile. It proved negative, with toxicities exceeding those of standard treatment. Despite this, patients are being treated based on an unproven paradigm. There are ethical questions regarding the promotion of this type of ‘personalised treatment’ in the absence of data that it will be of benefit and cost-effective. Patients’ expectations must be based on factual information. This approach should only be applied widely, if at all, when the results of additional randomised trials are available.
I also agree that a lot of new cancer drugs are coming on to the market, but there are questions about the evidence of benefit required for their approval, with many drugs struggling to meet the minimal criteria of efficacy defined by ESMO or ASCO.
Drugging genomic changes in cancer is not straightforward, despite your optimism. PI3KCA is an excellent example of a gene mutated in many cancers. Yet the Fathers of this field recently admitted that that the clinical results of PI3KCA inhibitors have fallen considerably short of their expectations. This struggle for efficacy is due in large part to the complex genetic evolution of tumours, which provides multiple subclonal drivers of cell proliferation and survival (see, for example, recent TracerX papers) that will thwart the effects of a single agent, or even combinations. The question is how do we respond to this emerging knowledge of extensive intra-tumour genetic heterogeneity?”
 Giuseppe Curigliano
Giuseppe Curigliano
“The SHIVA trial is not an appropriate example. The trial was designed more than 10 years ago, using a next generation sequencing technology that was very limited, and with limited access to cancer treatments. Take a look at the recent trial of larotractenib for use in child, adolescent and adult cancer patients whose tumours test positive for the TRK fusion (NEJM 2018, 378:731–739). The results showed the drug works across multiple tumour types, with a 78% response rate. That is impressive.
As for PI3KCA inhibitors, while it’s true the results so far show little benefit in solid tumours, we have yet to see the results relating to alpha-selective PI3KCA kinase inhibitors, which are a different class of agent.
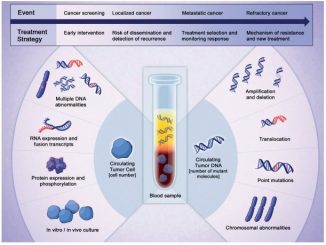
The answer to the problem of tumour heterogeneity and subclonal drivers is liquid biopsy. Liquid biopsies can identify, before clinical progression, subclones that are resistant to the treatment you are giving the patient, and enable you to select treatment accordingly. A good example is osimertinib, a targeted agent for non-small-cell lung cancer with the T790M mutation. The therapy is selected according to the mutation found in the liquid biopsy.
I anticipate that in future we will make the initial molecular diagnosis and select treatment using a liquid biopsy. The CancerSEEK test, which featured earlier this year in Science (18 January 2018), is a good example of how you can integrate several gene alterations and you can use this sort of technology not just for early detection but also to make the diagnosis.
As for the question about the level of benefit conferred by the many new cancer drugs, I don’t believe that precision medicine should have to improve survival to demonstrate that it is beneficial to the patient. Some of these agents improve survival in the adjuvant setting, some in the metastatic setting have an impact on progression free survival, which is I believe a valid endpoint, and also I believe response rate is valid in some cases.
What people in the metastatic setting are asking for is not duration of life but quality of life. Of course if you have improvement in survival I would be very happy, but in a setting in which you have nothing, an improvement in progression free survival is also an achievement”.
 John Hickman
John Hickman
“Increasing patient survival significantly is surely an important goal for the future. But, with an array of subclonal driver genes, the key question is what strategy is likely to succeed in doing this?
I don’t think liquid biopsy provides a solution to addressing tumour heterogeneity. I predict that advances in this technology will merely confirm the complexity of the challenge we face. So, yes, it may be used to confirm the emergence of mutations affecting a particular targeted therapy, as in the T790M mutation in EGFR, but it may also confirm the presence of multiple subclones with their own driver genes.
The majority of cancers are not monogenic diseases, with a dependence on one driver. They are unlikely to respond to any single agent the way chronic myelocytic leukaemia does. The presence of multiple subclones, such as are reported in the TracerX study of lung cancer, therefore remains the major challenge. I agree that some targeted therapies provide responses, like the TRK inhibitor you cite. Hopefully, in these particular cases, survival will also be extended significantly, but we should be careful about assuming the ALK (or TRK) paradigms will apply more generally.
Liquid biopsy could, however, play a key role in helping us move forward towards real cures, if we focus on developing its role in the setting of early detection. The CancerSEEK test you mention uses a mixture of genetic and protein biomarker tests to assess early signs of cancer. The results were somewhat variable, but the approach is admirable.
We now need to focus on harnessing our accumulating knowledge of the genetic changes initiating cancer. Precision cancer medicine with targeted therapies (together with other interventions) will have a greater chance to deliver increases in patient survival if cancers are treated early in their evolution, when heterogeneity is limited.
I would never question the value of developing treatments that improve the quality of life of people living with incurable cancer. What I question is whether the oncology community should be focusing so heavily on personalised treatment strategies that, on current evidence, are unlikely to deliver significant benefit for the majority of patients, and certainly not the major increases in survival that we need, nor at a price that allows the majority of patients access. A recent estimate suggests that only around 7% of patients benefit from genome-driven oncology (JAMA Oncol doi:10.1001/jamaoncol.2018.1660).
Oncologists have a responsibility, I believe, to pursue more promising avenues of research, in particular aimed at improving early detection. This should receive a much greater share of attention and effort”.
 Giuseppe Curigliano
Giuseppe Curigliano
“Targeted drugs for cancer date back to trastuzumab, approved for breast cancer in 1998, and the 2001 approval of the leukaemia drug imatinib, both of which have saved many lives. So far, the FDA has approved 31 targeted therapies for various cancers, including one that is approved for use against any cancer with a particular (DNA repair) mutation. Recent basket trials indicate that a similar approach could also work for other mutations, for instance, for cancers with TRK fusion. And although TRK fusions and the DNA repair mutations occur in only a small fraction of patients with a particular cancer type, when tallied across cancers, such drugs can help many patients.
As the list of targeted drugs grows, it makes sense to test tumours with genome-wide assays. If we don’t test people broadly, we will miss patients who have alterations for which there is now an approved therapy. In the trial that you mention, published in JAMA Oncology, 15.4% of 610,000 US patients with metastatic cancer were eligible for an FDA-approved, genome-guided drug. But they also found that, because the drugs shrink tumours in only some eligible patients, only 6.6% likely benefited. And many patients relapse after a couple of years on the drugs.
Analysis of current cancer genomic data sets suggests that we are still far from uncovering all the genetic drivers.
A major challenge for researchers working with cancer genomic data sets is their sheer size. The Cancer Genome Atlas data set alone is over a peta-byte in size, with more than 575,000 files. Just to download the data using a 10-Gbit-per-second connection would take over three weeks.
Setting up a secure, compliant infrastructure of sufficient scale to store and analyse the data is technically challenging and expensive. Artificial intelligence and the new generation computing equipment will help us match human genomic with clinical data. This will overcome the logistic and economic barriers by democratising access to cancer genomics data, enabling researchers to bring their hypotheses to the data.
Despite all the criticisms, we cannot stop advances in cancer research. On 25 May 2018, the European Union General Data Protection Regulation (GDPR) took effect. It may now be possible for individual patients to become ‘cancer information donors’, which would allow their genomic data to be shared through specific platforms. Mechanisms for enabling such donations are being developed under the GDPR. Given appropriate informed consent systems, we could identify patients with rare molecular subtypes of cancer who could be contacted for potential participation in clinical trials appropriate for their particular cancer.
Clearly, the principles and practice of precision oncology will be accelerated by sharing data from thousands of patients with cancer. In my opinion, genomic data will have a pivotal role in precision oncology. A worthy goal will be to develop a new taxonomy of disease based on molecular pathogenesis and to demonstrate it has clinical utility in cancer treatment.
The future will be facilitating the sharing of cancer genomic and clinical data”.