The impact cancer and cancer treatment can have on a persons sex life can cause physical, emotional and relationship difficulties. This overview describes the many problems that can be triggered, and a step-wise approach to addressing them, with a special focus on step one: asking patients about their sexual concerns.
 Sexual difficulties can affect everyone at some point in their lives and we need to be aware of the rates of sexual dysfunction reported in healthy populations when considering how cancer might cause sexual difficulties in addition to those occurring generally. A cross-sectional survey of healthy US adults aged 18–59 found that 31% of men and 43% of women reported some degree of sexual difficulty. Ten per cent of men had difficulty achieving or maintaining an erection, 15% had low sexual desire, 8% of men couldn’t reach orgasm and 30% of men experienced rapid ejaculation. In women, 32% reported a lack of interest in sex, around 14% had arousal difficulties and 7% of women reported sexual pain (J Sex Med 2008, 5:289–300). Estimates from a range of studies suggest about 25% of women have difficulty achieving orgasm. Age is a major risk factor for increased rates of sexual difficulties in both men and women.
Sexual difficulties can affect everyone at some point in their lives and we need to be aware of the rates of sexual dysfunction reported in healthy populations when considering how cancer might cause sexual difficulties in addition to those occurring generally. A cross-sectional survey of healthy US adults aged 18–59 found that 31% of men and 43% of women reported some degree of sexual difficulty. Ten per cent of men had difficulty achieving or maintaining an erection, 15% had low sexual desire, 8% of men couldn’t reach orgasm and 30% of men experienced rapid ejaculation. In women, 32% reported a lack of interest in sex, around 14% had arousal difficulties and 7% of women reported sexual pain (J Sex Med 2008, 5:289–300). Estimates from a range of studies suggest about 25% of women have difficulty achieving orgasm. Age is a major risk factor for increased rates of sexual difficulties in both men and women.
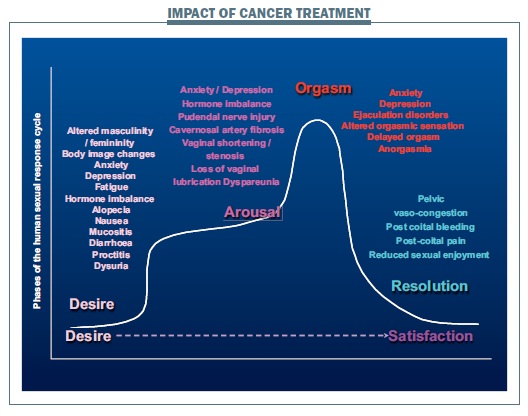
Impact of cancer treatment on phases of the human sexual response cycle
Cancer treatment can affect all phases of the human sexual response cycle.
Desire phase
The ability to feel desire and to feel that others might desire you sexually can be affected by body image changes, altered masculinity and femininity caused by treatment, and anxiety or depression associated with being diagnosed with cancer. Sexual interest is also affected by fatigue as a consequence of radiotherapy and chemotherapy, changes in hormones as a direct result of cancer therapy – particularly after breast, gynaecological or prostate cancer, and treatment side-effects including loss of hair, feeling nauseated, problems with diarrhoea and mucositis (painful inflammation or ulceration in the mouth or digestive tract).
Arousal phase
During the arousal phase, a man’s ability to get and keep an erection can be indirectly affected by changes in testosterone and directly affected by nerve injury to the pudendal nerve or through scarring and fibrosis of the cavernosal arteries caused by radiotherapy or by surgery. In women, changes to the vagina in terms of shortening or narrowing caused by surgery or pelvic radiotherapy and loss of vaginal lubrication can lead to sexual pain and reduce the ability to relax and enjoy arousal.
Orgasm
Anxiety and depression and drug treatments for them can have a negative impact on the ability to reach the threshold necessary to have an orgasm. Even when an orgasm is achieved, it may be less intense and less enjoyable. Nerve damage can also cause problems with ejaculation. Men may fail to ejaculate at all or may have retrograde ejaculation, where the ejaculate passes back into the bladder, rather than being expelled forwards out of the end of the penis. There can also be delays in being able to achieve an orgasm, or loss of orgasm altogether.
Question: How can you help women with reduced sensation after pelvic cancer to reach orgasm?
Answer: The first point is to ensure we ask the question about whether any aspect of a woman’s sexual wellbeing has been adversely affected by her cancer treatment. If she reports a change in orgasm, there are a variety of techniques and approaches that might be suggested, usually by someone trained as a sex therapist or a psychologist or a nurse with training in cognitive behavioural approaches for sex therapy. We would usually use a technique called sensate focus, which helps women, through a series of exercises carried out at home with her partner, to identify different erogenous zones and what types of stimulation and touch are enjoyable. She might also consider using a lubricant that can help stimulate sensation or a vibrator, which may help orgasm where stimulation by oral sex or fingers might not be sufficient.
 Prevalence of sexual difficulties after cancer
Prevalence of sexual difficulties after cancer
The prevalence of sexual difficulties is generally higher after cancer than in the general population. A survey by Macmillan Cancer Support, “Worried Sick: the Emotional Impact of Cancer”, found that 43% of UK patients said that their sex life had suffered as a direct consequence of their cancer treatment. The rate of sexual difficulties is higher – 50% to 80% – in patients who’ve had pelvic radiotherapy for cervical cancer (BMJ 1994, 308:869–870; Int J Radiat Oncol Biol Phys 1995, 31:399–404). After breast cancer, particularly after treatment-induced menopause, the prevalence of sexual difficulties can be as high as 70% (J Sex Med 2010, 8:294–302). The rate is similarly high (70%) after prostate cancer, irrespective of whether treated surgically or by radiotherapy and hormone therapy (Int J Radiat Oncol Biol Phys 2006, 66:439–444; Sexologies 2007, 16:273e278). After rectal cancer treatment 86% of men report sexual difficulties (J Sex Med 2010, 7:349–373), but there are limited data for women with rectal cancer.
These figures show that sexual difficulties are common after cancer. But not all patients necessarily want to pursue treatment for these problems, and it’s part of the skill of clinicians to find out whether sexual recovery is something that’s important to a particular patient or couple.
We automatically think of patients with cancers that pose high risk of sexual difficulties, but we also need to consider lower profile patient groups. These include patients treated for haematological cancers, particularly those treated with stem cells or bone marrow transplantation, who often need advice on how to prevent infection, how to reduce the risk of bleeding associated with sexual activity during treatment and where there might be difficulties with vaginal or penile graft versus host disease. Other patient groups that are also neglected include those treated for head and neck cancer, cerebral tumours, people who have had amputations or limb-preserving surgery that affects their mobility and ability to adopt certain sexual positions and teenagers and young adults, where research is lacking.
Question: We know a lot about how surgery and radiotherapy affects sexual functioning in men and women but what about chemotherapy?
Answer: The effects are mainly to do with whether or not it’s safe to have sexual activity during periods where patients might be immunosuppressed or where their thrombocyte count is significantly reduced. Generally, the sort of advice that we might give would be to suggest that barrier contraceptives can reduce the risk of sexually transmitted infections while someone is immunosuppressed, as well as protecting against unplanned pregnancy. Patients also worry about whether they may bleed more heavily, associated with chemotherapy-induced thrombocytopenia, and advice may include using a good lubricant to reduce friction and being gentle during penetrative sex.
The other area that is sometimes asked about is anal sex – and it’s not just gay male couples that engage in anal sex, heterosexual couples may include it in their sexual repertoire. Patients who are immunosuppressed or thrombocytopenic are advised not to practice rectal penetration because the risk of infection is higher with anal sex. Rectal or anal intercourse can be dangerous in patients who are thrombocytopenic because there is a greater risk of mucosal damage and bleeding.
Other chemotherapy-associated problems relate to complications such as fatigue. Patients ability to enjoy sexual contact might be reduced during the immediate post-treatment period when fatigue is most severe.
Cancer and sex: myth or reality?
There are lots of myths that seem to surround cancer and sexual activity. These myths are often not volunteered and may need to be skilfully brought into the conversation to normalise their existence in order to correct misconceptions. A common myth is the fear of contamination from radioactivity. Patients who have had pelvic radiotherapy may worry that they can pass radiation on to their partners. But there is no possibility of patients treated with external beam radiotherapy passing on radioactivity. The only situation where you would recommend avoiding sexual contact would be in a patient treated with sealed or unsealed sources such as iodine-131 who may still be considered radioactive.
Some patients worry about cytotoxic contamination of their partner. There is a theoretical fear about being contaminated by the breakdown products of chemotherapy agents, but there is no research to indicate that this has ever happened, and I’ve never come across male or female patients whose partners developed irritation or difficulty associated with sexual contact. Patients who are very nervous about this might be advised to use a barrier contraceptive, but there is no theoretical or research evidence to back up this advice.
Some patients worry about whether their cancer can be passed on to their partner and can be reassured that this is not possible. Patients on treatment to suppress hormone levels, such as in prostate or breast cancer, can worry that having sex might lead to a surge in oestrogen or testosterone levels, but there is no evidence to suggest that this happens so we advise that sexual activity is perfectly safe.
A more common fear is that sex might cause pain or damage. Patients can be reassured that it’s quite safe to have gentle sex during or after treatment for cancer. Some patients worry that sexual contact in the past has caused their cancer. For human papillomavirus associated cancers, it is helpful to explain that most sexually active adults are exposed to HPV, but not all of them contract a cancer that is associated with that exposure. For HIV-related cancers, advice should include safe sex messages with the use of appropriate barrier contraceptive methods to reduce the likelihood of HIV transmission.
Some patients worry about being re-infected after successful treatment for a virally associated cancer if they believe their partner is the source of the virus. This often requires a conversation between the patient, their treatment team (maybe with advice from a virologist) and a sexual counsellor, to work through the period of fear or anxiety.
Asking patients about sexual concerns
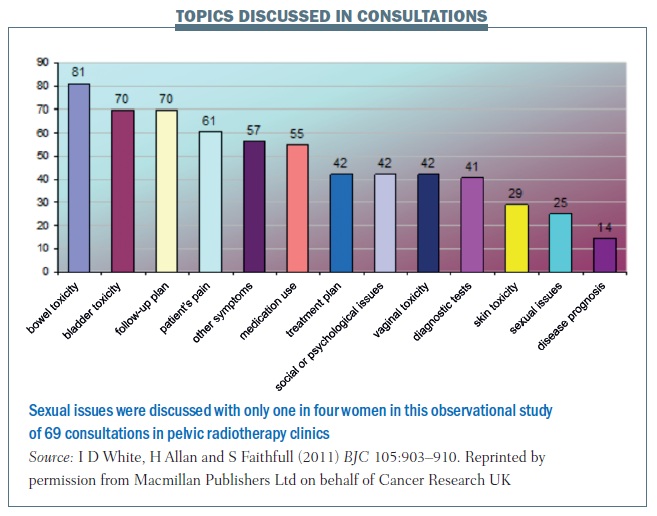
Clinicians are reluctant to ask cancer patients about sex during reviews in outpatient or follow-up clinics. A study I carried out in pelvic radiotherapy clinics for women showed that sexual issues were discussed in only 25% of consultations – even though this is a high-risk group for sexual difficulty – while bowel toxicity was discussed in 81% and bladder toxicity in 70% of consultations (see figure below).
In men undergoing treatment for prostate cancer, the prevalence of inquiry about sexual difficulties was 52% in radiotherapy clinics and 54% in urology clinics (Br J Urol Int 2011, 109:98–103). Questions about sexual recovery were generally raised by healthcare professionals. They asked about sexual concerns in 48% of radiotherapy consultations and 30% of urology consultations observed (figures for patients were 4% and 22%, respectively). However, even though this is a high-risk group of patients, the question of sexual recovery wasn’t addressed in around half of consultations, although this is better than the 25% figure in female patients.
It is important to recognise that the majority of people whose sexual lives are affected by cancer treatment can benefit from timely accurate information given by their treatment team regarding the common sexual consequences they may encounter and where to seek support/information if they have concerns. Some may benefit from brief sexual counselling or more detailed information, given by advanced practitioners or nurse specialists in oncology or sexual health. Those likely to require psychosexual therapy or specialist counselling are usually a small number of individuals with more persistent or complex sexual difficulties and high levels of individual or couple distress.
Question: Could you give an example of a question that you could ask a patient when you start to talk about sex?
Answer: You need to help the patient to feel as though it is alright to ask a question, so I say things like: “Many patients who have had your kind of treatment (or cancer) have fears or worries about what it might feel like to get back into sexual activity again afterwards. Is that something that is worrying you?”
For research purposes, it’s helpful to use established instruments such as the International Index of Erectile Function and the Female Sexual Function Index to assess sexual functioning to provide consistency when you’re reviewing patients at regular intervals during follow-up. Many of the EORTC quality of life instruments, including those for breast, cervical, prostate, endometrial and rectal cancer, have a small number of questions on sexual function. The EORTC is currently developing male and female modules specifically addressing sexual recovery or adjustment after cancer.
 Managing sexual difficulties
Managing sexual difficulties
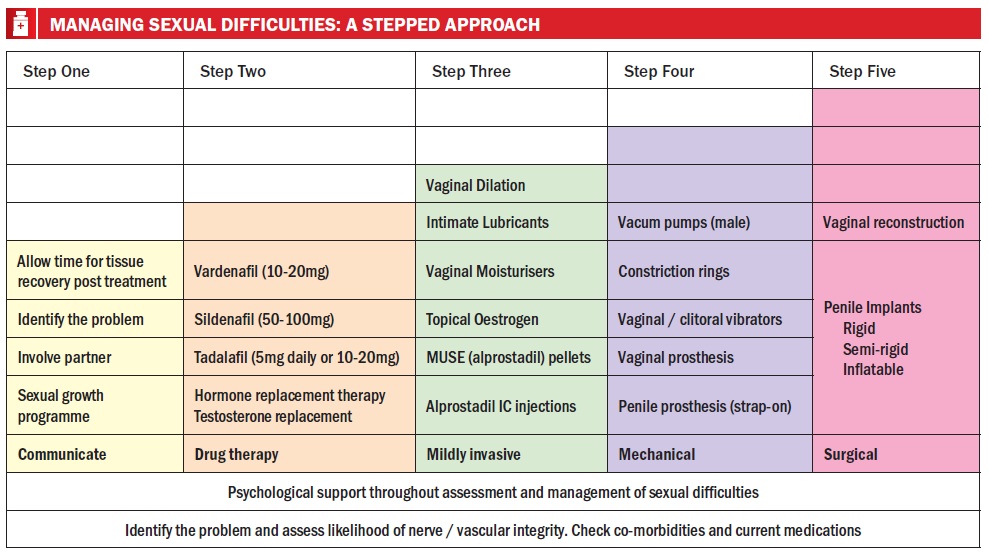
The figure below shows a stepwise approach to sexual difficulties caused by cancer or cancer treatments.
Step 1: The initial step is to identify the problems through assessment, involving the partner where that is appropriate, and helping the patient communicate their difficulties and anxieties about sexual recovery.
Step 2: If we identify sexual difficulties, step two is to consider drug treatment, including drugs for erectile dysfunction or hormone replacement therapy for someone with a treatment-induced menopause or testosterone deficiency that’s adversely affecting their sexual function, as long as it’s oncologically safe to do so.
Step 3: We might also consider step three, including advising patients on the use of vaginal dilators if they’ve had either pelvic radiotherapy or pelvic surgery, particularly where they may have had vaginal reconstruction. (International Guidelines on Vaginal Dilation after Pelvic Radiotherapy can be found on the National Cancer Survivorship website www.ncsi.org.uk). We also commonly suggest the use of intimate lubricants, vaginal moisturisers or topical oestrogens, to help with treatment-induced vaginal changes. For men, we may use alprostadil for erectile dysfunction not responding to oral therapy.
Step 4: Step four provides mechanical options, including vacuum pumps, constriction rings, and vaginal or clitoral vibrators for orgasmic difficulties. We might consider a vaginal prosthesis for a woman without a vagina who wants to have penetrative sex, or a penile prosthesis (strap-on) for a male patient who cannot maintain an erection by other means.
Step 5: Lastly, step five is surgical interventions, with vaginal reconstruction techniques or penile implants for patients where other approaches have not been successful.
Psychological support, either through psychotherapy, sexual counselling or psychosexual therapy, should be available throughout assessment and management. Psychosexual therapy can be useful for a variety of sexual difficulties, including persistent loss of sexual desire, orgasmic difficulties, or for patients who are fearful of having lost femininity/masculinity. Strategies helping patients to adjust to non-coital alternatives can be used where biomedical treatments for erectile dysfunction have not worked. A couples therapy approach would be preferred where both people in a couple are distressed.
A Cochrane review on interventions for sexual dysfunction following cancer treatment (Interventions for sexual dysfunction following treatments for cancer, Issue 4. Art. No.: CD005540. doi: 10.1002/14651858.CD005540.pub2) found the strongest evidence for oral phosphodiesterase 5 inhibitors for erectile dysfunction after radical prostatectomy or radiotherapy. Only one of the studies reviewed explored the effectiveness of a vaginal cream containing oestrogen after radiotherapy for cervical cancer, and found some evidence for efficacy. More research is needed in this field.
Question: Women with pelvic cancers such as gynaecological cancers and rectal cancer are afraid of topical oestrogen. Is that your experience as well?
Answer: Yes. For women with cervical cancer or rectal or anal cancer, I usually explain that as their cancers are not hormone responsive we don’t think that there are any safety concerns with topical oestrogen. The only women who should not use vaginal oestrogen are those who’ve been treated for endometrial cancer, or women treated for breast cancer where their consultants consider it not advisable.
 Psychosexual therapy
Psychosexual therapy
Sex therapists work across three domains of sexual function:
Physical function, such as hormonal, vascular, and neurological changes, other physical illnesses and drug therapy that might be causing problems with sexual recovery,
Psychological response, including anxiety, depression and other concurrent mental health difficulties that might make psychological wellbeing a challenge.
Couple interaction. Not all couple relationships are supportive and there might be fear, blame, anger or a history of abuse requiring further psychosexual therapy and support.
We look at three sets of factors when assessing patients with sexual difficulties that they can’t overcome without help. These include predisposing factors for being unable to adjust to sexual changes, such as previous abuse; precipitating factors, such as the cancer diagnosis and treatment, or relationship breakdown; and factors that maintain difficulties, such as poor couple communication or fear and anxiety in one or both members of the couple.
A retrospective cohort study of 259 women attending a survivorship programme after a variety of cancers (37% gynaecological, 92% postmenopausal) found the commonest sexual problems were dyspareunia (painful intercourse), atrophic vaginitis (changes to the skin and lubrication of the vagina walls that can result in the vagina becoming shorter, less elastic and drier), and decreased sexual desire. Orgasmic difficulties occurred in a minority of patients (J Sex Med 2006, 3: 646–649).
Treatment recommendations inclu-ded hormone therapy (89%), psychosexual counselling (46%), vaginal oestrogens (34%), and vaginal dilators (25%). There was 63% compliance at six months and 70% of the women reported a subjective improvement in their symptoms. A literature review suggested moderate support for the effectiveness and the feasibility of psychological interventions for sexual dysfunction after cancer and cancer treatments (J Cancer Survivorship 2010, 4:346–360) I think that this shows that, with increased awareness, there are more patients that we can help with approaches that go beyond drug-focused interventions for sexual difficulties after cancer and cancer treatment.