Screening breast cancer patients for lymphoedema makes it possible to identify problems early and take action to halt its progression. Oncologists have an important role to play, particularly given the rise in lymphoedema rates that may be expected due to greater use of regional radiotherapy in early breast cancer.
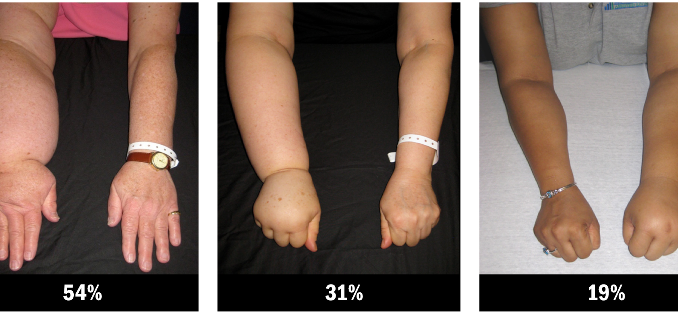
 In the past, lymphoedema was relatively common in women following treatment for breast cancer. It could be severe, with arms swelling up to one and a half times their normal size, which is referred to as 50% lymphoedema. Thankfully it is rare to encounter problems of this scale today.
In the past, lymphoedema was relatively common in women following treatment for breast cancer. It could be severe, with arms swelling up to one and a half times their normal size, which is referred to as 50% lymphoedema. Thankfully it is rare to encounter problems of this scale today.
The aim of this review is to change the way we think about breast cancer-associated lymphoedema.
I would like to propose that we detect swelling early, at a level of around 7–10%, which increases the possibility of treating it successfully.
How do we think about lymphoedema today? We think about it in terms of an impairment-based model. This means that when we see lymphoedema, we start to treat it. Treatment is generally unsuccessful in more severe cases, and physicians tend not to be very involved. As soon as we see swelling we just send the patient to a physical therapist to provide massage or other therapy, but the success rate is fairly low. Limitations hampering improved management are that we do not have a universal definition of lymphoedema or an accurate method to measure it, and there is no level 1 evidence – no phase III randomised trial – on the best way to treat lymphoedema.
For the future, I would like to change the model for lymph-oedema to a screening-based model in which we no longer wait until we see that a patient’s arm has swollen but, instead, we screen for the problem, detect it early on and then test the best way to treat it. Oncologists should take a lead in this approach because we see the patient very early on during their treatment, while a physical therapist sees them once the problem is already there.
We need to come together to define lymphoedema, agree what degree of swelling we call lymphoedema, and the optimal method to measure it. We need to determine when we should intervene: should it be when there is minimal swelling, a 3–5% difference in volume for example, or 5–10% or more than 10%? We also need to generate level 1 evidence with phase III randomised trials to determine the standard of treatment. And we need to be very mindful of the cost of the treatment.
Our department has been interested in lymphoedema for some time and considers that the only way to move this field forward is to work as a team. The Massachusetts General Hospital lymphoedema team, initially formed in 2005, includes: a breast radiation oncologist, physical therapist, surgical oncologist, clinical research coordinator and a patient advocate, all of whom came together to think how best we can address lymphoedema. The goals are to:
- identify lymphoedema as early as possible
- empower patients to manage lymphoedema with as little burden as possible, keeping their goals central to decision making
- contribute to the scientific literature by generating level 1 evidence
- see if early detection leads to early intervention and whether this results in better outcomes.
What do we know about lymphoedema?
The incidence of clinical oedema is 20–25% in patients undergoing axillary node dissection, and 5–9% in those undergoing sentinel node mapping. Subclinical oedema is experienced by almost half of patients with axillary node dissection (47%) and by 15% of patients undergoing sentinel node mapping. There is no doubt that improving surgical technique has greatly reduced the risk of lymphoedema, although it is still high in regions that use lymph node dissection.
Established risk factors include: axillary dissection, wound infection, axillary radiation and high body mass index (BMI). A BMI of 30 or more greatly increases the risk (Breast Cancer Res Treat 2013, 142:59–67). Potential risk factors include the number of nodes removed (although this has not been found in patients undergoing sentinel lymph node biopsy alone- (Ann Surg Oncol 2010, 17:3278–86) and the number of positive nodes (JCO 2008, 26:3536–42).
The negative impact of lymph-oedema on patients’ quality of life is very well established. It can also have a negative impact on body image, with the patient having to live with a permanently swollen arm constantly reminding her about her breast cancer. When lymphoedema is significant, it can decrease the upper extremity function, which also impairs quality of life.
Several studies show that exercise is good for lymphoedema and does not exacerbate the problem (NEJM 2009, 361:664–673; Breast Cancer Res Treat 2008, 109:9–26; Biol Res Nurs 2008, 10:34–43). There are a lot of myths in the lymphoedema field, such as that repetitive arm movements can generate lymph and compromise lymph drainage and can cause more swelling. These still appear in the brochures that we give to patients, but we now know it is completely wrong. The data support exercise, in particular weight lifting, carried out in a well-controlled and progressive manner.
What is the best method to quantify changes in arm volume?
There are four different techniques in the literature:
Tape measurement. The most popular way of measuring lymph-oedema is by measuring the circumference of the arm with a tape measure (Cancer Investigation 2005, 1:76–83). Unfortunately, physicians are not usually very thorough, measuring only one or two points of the arm, such as 10 cm above or below the elbow. This method has poor reliability and is unable to quantify swelling in the hand or the breast. A more thorough approach is to measure the circumference of the arm every 4 cm, and use software to generate the volume of the arm.
Water displacement. This is the second most commonly used way to measure lymphoedema, and requires the patient to put her arm in a large glass cylinder. The water displaced reflects the arm volume. This method is commonly used in clinical trials; however, it is messy and unhygienic, it takes time, and the reliability is questionable.
Bioimpedance. This relatively new method was developed in Australia. It calculates an impedance value, which reflects the ability of an electrical current to pass through the limb. It is quick and very convenient, simply requiring a small machine that can easily be taken from one consultation room to another. However, as it is rather new there are limited data.
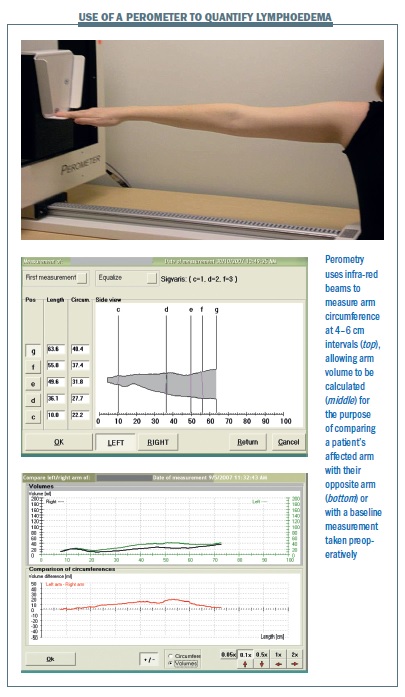
Perometer. This equipment uses infra-red technology to quantify lymphoedema. The patient extends her arm horizontally while a frame moves back and forth producing an image of the volume of the arm on a computer screen (see figure overleaf). This can be compared with the other arm, or baseline measurements, to give the percentage of swelling. We are currently using this method in our lymphoedema screening programme. It is convenient and accurate but the equipment has to be installed into a dedicated room.
 What is the definition of lymphoedema?
What is the definition of lymphoedema?
The consensus in the literature, which we use in our programme, defines lymphoedema as a 10% increase in volume compared to the arm volume in the non-treated side. However, several other definitions are in current use, which I believe are less useful.
For instance, lymphoedema is often reported as >2 cm difference in circumference by tape measure at a certain location in the arm (Lymphat Res Biol 2005, 3:208–217), or as a volume increase of 150–250 ml Mayo Clin Proc 2005, 80:1480–84). Both of these are absolute values, which is unhelpful because they don’t take into account the wide variation in normal arm size. A patient with thin arms who shows a volume increase of 180 ml will not meet the definition of 200 ml increase even though her arm has swollen to 15% larger than the opposite arm. She will not be diagnosed as having lymphoedema so will miss out on treatment. A patient with a very large arm, on the other hand, may show a swelling of 220 ml, so meeting the definition of lymphoedema, even if this represents a difference of only 3.5%. This patient might therefore be overtreated (Breast Cancer Res Treat 2012, 135:145–152).
Defining lymphoedema on the basis of a percentage increase in volume is therefore more useful. Various thresholds have been suggested (see below); the consensus in the literature is on 10%, which is what we use.
How do we calculate the volume of lymphoedema?
One important thing that many people forget is that at least one in five women have one arm larger than the other; and one in 20 have a volume difference of as much as 10% at baseline. If you don’t have this information upfront, you might over- or under-estimate the level of lymphoedema. We studied this in 677 consecutive patients undergoing unilateral breast cancer surgery, measuring relative (percentage) volume change in the at-risk arm compared to pre-operative baseline, using the other arm as a control. The results generated the following relative volume change (RVC) formula:
RVC = (A2U1)/(A1U2) – 1
(where A1, A2 are at-risk arm volumes at pre-op baseline and post-op follow-up, and U1, U2 are arm volumes on the contralateral side at corresponding times) (Int J Radiat Oncol Biol Phys 2011, 79:1436–43).
In bilateral breast cancer, there is no control arm, and both sides have swelling, so we measured the relative change in at-risk arm volume compared to the pre-operative baseline in a study of 265 unilateral surgery patients and obtained the following weight-adjusted volume change (WAC formula):
WAC=(A2W1)/(A1W2) – 1
(where A1, A2 are at-risk arm volumes at pre-op baseline and post-op follow-up, and W1, W2 are the patient’s weight at corresponding times) (Lymphology 2013, 46:64–74).
 What is the appropriate threshold to initiate intervention?
What is the appropriate threshold to initiate intervention?
The aim is to intervene at an early stage, before the patient’s arm becomes very enlarged. Should we intervene at 3%, 5%, 10% or 20%? We don’t yet know.
Based on symptoms, different investigators have recommended intervention at 10% volume difference (Palliat Med 2005, 19:300–313); 20% (Eur J Cancer 2003, 39: 2165–67); 200 ml (Breast Cancer Res Treat 2002, 75:51–64); 250 ml (Am J Surg 1999, 178:311–315); or 2 cm circumference difference. These are all a bit too high. The NIH study, which included only 43 patients, intervened at more than 3% volume increase (Cancer 2008, 112:2809–19). However, we consider this too small because a volume increase of 3–5% might represent temporary swelling after surgery.
We looked at the best threshold to intervene in a study of the natural history of almost 1500 patients, carrying out repeat measurements and following them over time. We did not intervene in patients with less than 10% difference. We found that women who had 5–10% swelling, some with very minimal, sub-clinical swelling, progressed to have more severe lymphoedema. Most patients with a volume increase of less than 3% recovered. This suggests the threshold for intervention could be a 5–10% volume increase, but a trial is needed to test whether intervention in these patients is beneficial or not. The trial should randomise patients with a volume increase within this range to observation or intervention (a simple sleeve).
It is important to remember that transient lymphoedema, which resolves without intervention, is common. A trial of 918 patients showed transient lymphoedema in 71% of patients, with a relative volume change of 5% or more for at least three months. It was persistent in the remaining 29% (J O’Toole et al, Congress of Lymphology, International Society of Lymphoedema, 19-23 September 2011).
Why is lymphoedema so important today?
Two very big studies, one from Canada and one from Europe, showed that women who have a small number of positive lymph nodes (1–3 positive lymph nodes) or high-risk node negatives benefit from regional radiation (T Whelan et al, ASCO 2011 Abstract LBA 1003), but this increases the risk of lymphoedema.
We can therefore expect regional radiation to be used more often, which is likely to increase the rate of cases of lymphoedema over the next 5–10 years.
The risk factors for lymphoedema (defined as an increase in volume of ≥10%) that we have found from analysing our data are:
Measured arm volume changes, (where RVC = relative volume change):
RVC 3% – <10% in the first 3 months after surgery indicates high risk of lymphoedema
RVC 5 – <10% any time after 3 months post-surgery indicates possible risk of developing lymphoedema.
Clinical and treatment-associated factors:
- BMI ≥30 at the time of surgery
- Axial lymph node dissection
- Regional lymph node radiation
Overall, one in every four or five patients with these risk factors will develop lymphoedema.
What is the appropriate treatment for low-volume lymphoedema?
The question for the future is whether early intervention will prevent progression to more severe lymphoedema. One of the ideas we are considering is a study randomising patients with early swelling to three groups:
- a control group who receive the standard management, such as counseling and stretching but no specific treatment, and are observed to see whether lymph-oedema develops
- intervention with a compression sleeve, worn for at least 12 hours a day for 12 weeks, or
- interventional exercise to stop lymphoedema from progressing, including cardiovascular exercise and progressive weight lifting (30 minutes, 3–4 times each week for 12 weeks).
 Conclusions
Conclusions
We have to change the way we manage lymphoedema. It is not acceptable to wait until a patient develops a largeswollen arm, because of the negative impact this has on the quality of her life for the rest of her life.
Sleeves are not curative; they can only provide a control measure to help mitigate the ongoing problem.
Instead, we need to screen for lymphoedema, define it early and then intervene with the aim of preventing progression. There is a great need to study early intervention to generate level I evidence, such as conducting a randomised study.